Chemistry - Online Test
Q1. Which among the following is an example of pseudo first order reaction?
Answer : Option C
Explaination / Solution:
In acid catalysed hydrolysis of ethylacetate the water is in excess so it is pseudo 1st order reaction, as cincentraton does not change.
Q2. When adsorption of oxalic acid is carried out on activated charcoal, then activated charcoal is known as
Answer : Option C
Explaination / Solution:
Surface on which adsorption occurs is known as adsorbent.
Q3. 34.05 mL of phosphorus vapour weighs 0.0625 g at 546 and 0.1 bar pressure. What is the molar mass of phosphorus?
Answer : Option B
Explaination / Solution:
PV=(0.0625/M)RT where p=0.1bar T=819.15K and V= 34.05ml
Q4. The enzyme which catalyses the hydrolysis of fats into fatty acids and glycerol is.
Answer : Option C
Explaination / Solution:
Lipase catalyse hydrolysis of fats into fatty acids and glycerol.
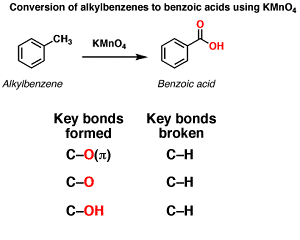
Q5. The compound formed as a result of oxidation of ethyl benzene by KMnO4 is
Answer : Option B
Explaination / Solution:
Oxidation of aromatic alkanes with KMnO4 to give carboxylic acids. Description: Treatment of an alkylbenzene with potassium permanganate results in oxidation to give the benzoic acid.
Q6. In which of the following solvents is silver chloride most soluble?
Answer : Option A
Explaination / Solution:
AgCl is soluble in ammonia due to the formation of complex
Q7. In pressure cooker boiling point of water
Answer : Option A
Explaination / Solution:
As pressure increases boiling point also increase.
Q8. Arrange the following compounds in order of increasing boiling point:
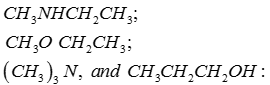
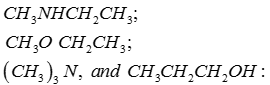
Answer : Option C
Explaination / Solution:
This is on the basis of inter molecular interactions.
Q9. An oxidation number of +1 is found in all their compounds of one of the below given options
Answer : Option B
Explaination / Solution:
The alkali metals (Group 1) have 1 valence electron. Alkali metals looses this electron to achieve noble gas configuration, and so alkali metals have oxidation number +1.
Q10.
Given that Avagadro’s Number
= 6.02 x 1023 atoms/mol ,
the number of hydrogen atoms in 18g of water is.
Answer : Option B
Explaination / Solution:
Since,18g H2O
= 1mol water containing 6.02 x 1023 molecules of water (H2O )
& the number of H atoms in 1 molecule of hydrogen (H2) = 2
the number of H atoms in 6.02 x 1023 molecules
= (6.02 x 1023 x 2)
=1.204 x 1024.
