Chemistry - Online Test
Q1. Which colour is given by CuSO4 with ammonia?
Answer : Option A
Explaination / Solution:
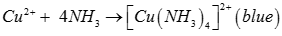
Q2. Among the following compounds the one that is most reactive towards electrophilic nitration is
Answer : Option A
Explaination / Solution:
Methyl group is electron donating group, hence it increases the electron density in benzene ring thereby increasing the reactivity of the ring towards electrophilic substitution.
Q3. Which among the following act as flux , if iron oxide is present as an impurity?
Answer : Option D
Explaination / Solution:
Iron oxide is basic oxide so flux should be acidic.
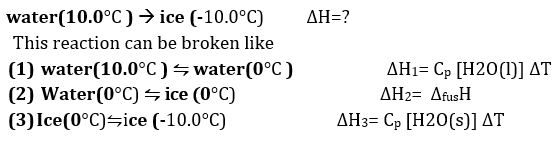
Q4. Calculate the enthalpy change on freezing of 1.0 mol of water at 10.0 to ice at -10.0. H = 6.03 kJ at 0.1
Answer : Option C
Explaination / Solution:

Q5. Which among the following is an example of non – polar molecule?
Answer : Option A
Explaination / Solution:
Methane is a symmetrical.
Q6. Right order of increasing metallic character is:
Answer : Option A
Explaination / Solution:
Elements of s block are more matallic than p block elements
Q7. Which of the following polymer can be formed by using the monomer unit represented below?
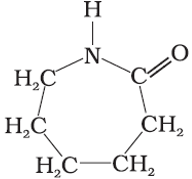

Answer : Option D
Explaination / Solution:
Nylon – 6, 6 Caprolactum is monomer of nylon 6.
Q8.
The electronic configuration belongs to
Answer : Option A
Explaination / Solution:
Boron is a chemical element with symbol B and atomic number 5. So electronic configuration of boron is
Q9. Which of these atmospheric pollutants is not released by car exhausts ?
Answer : Option D
Explaination / Solution:
Magnesium oxide is not released by car exhaust because Mg is not present in the fuels.
Q10. The production of oxidation of I– withMnO4−− in acidic medium is
Answer : Option C
Explaination / Solution:
In acidic medium I− oxidizes to I2
