UNIT 8: Physical and Chemical Equilibrium - Online Test
Kb = 0.8 × 10–5
Kf = 1.6 × 10–4
Keq = Kf / Kb = 1.6X10-4
/ 0.8X10-5 = 20
At a given temperature and pressure, the equilibrium
constant values for the equilibria
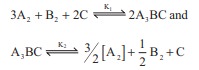
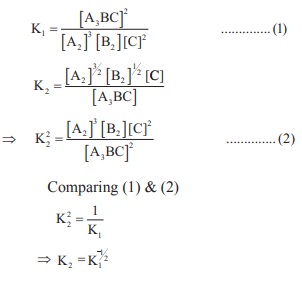
T1 = 25 + 273 = 298 K
T2 = 700 K

ΔHº is –ve ie., forward reaction is exothermic
The formation of ammonia from N2(g) and H2(g)
is a reversible reaction
N2(g) + 3H2(g) ⇌ 2NH3(g) + Heat
Increase in temperature, favours the endothermic reaction,
Given that formation of NH3 is exothermic ie.,
the reverse reaction is endothermic.
∴ increase
in temperature, shift the equilibrium to left option (c)
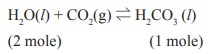
option (a) : wrong statement
Correct statement is, for a system at equilibrium, Q = Keq
K1 and K2 are the equilibrium
constants for the reactions respectively.
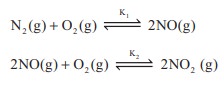
What is the equilibrium constant for the reaction NO2(g)
⇌ ½N2(g) + O2(g)

In the equilibrium,
2A(g) ⇌ 2B(g) + C2(g)
the equilibrium concentrations of A, B and C2 at
400 K are 1 × 10–4 M, 2.0 × 10–3 M, 1.5 × 10–4
M respectively. The value of Kc for the equilibrium at 400 K is
[A] = 1 × 10–4
M ;
[B] = 2 × 10–3 M
[C] = 1.5 × 10–4 M
2A (g) ⇌ 2B(g) + C2(g)
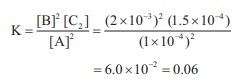
Kc = [Products] / [Reactants]
3.2 x 10-6 = [Products] / [Reactants]
KC < 10-3 ; indicates
that [Reactant] >> [Product]
option (b) is correct,largely towards reverse direction.
for the reaction,
N2(g) + 3H2(g) ⇌ 2NH3(g) ;
Δng = 2 – 4 = – 2
∴ KP
= KC (RT)–2
Kc/Kp = (RT)2
