UNIT 5: Alkali and Alkaline Earth Metals - Online Test
Q1. For
alkali metals, which one of the following trends is incorrect ?
Answer : Option C
Explaination / Solution:
Potassium is lighter than sodium (Refer table 5.3)
The correct order of density is Li < K < Na
< Rb < Cs
Q2. Which
of the following statements is incorrect ?
Answer : Option A
Explaination / Solution:
Li+ has maximum degree of hydration
among alkali metal cations.
Li+ > Na+ > K+
> Rb+ > Cs+
Q3. Which
of the following compounds will not evolve H2 gas on reaction with
alkali metals ?
Answer : Option D
Explaination / Solution:
All
these compounds reacts with alkali metals to evolve hydrogen gas.
Q4. Which
of the following has the highest tendency to give the reaction M+(g) --Aqueous
---Medium → M+(aq)
Answer : Option B
Explaination / Solution:
hydration
energy of Li+ is more and hence Li+ is stabilized in
aqueous medium.
Q5. Sodium
is stored in
Answer : Option C
Explaination / Solution:
No Explaination.
Q6. RbO2
is
Answer : Option A
Explaination / Solution:
RbO2
is a super oxide which contains Rb+ and O2– ions. O2–
contains one unpaired electron and hence it is paramagnetic.
Q7. Find
the wrong statement
Answer : Option C
Explaination / Solution:
Potassium carbonate cannot be prepared by solvay
process. Potassium bicarbonate is fairly soluble in water and does not
precipitate out.
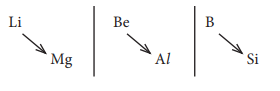
Q8. Lithium
shows diagonal relationship with
Answer : Option B
Explaination / Solution:
Q9. Incase
of alkali metal halides, the ionic character increases in the order
Answer : Option B
Explaination / Solution:
ionic character (difference in electronegativity)
MI < MBr < MCl < MF
Q10. In
which process, fused sodium hydroxide is electrolysed for extraction of sodium
?
Answer : Option A
Explaination / Solution:
Castner's process
NaOH ↔ Na+ + OH–
Cathode : Na+ + e– → Na
Anode : 2OH– → H2O + 1/2O2
+ 2e–
