Organic compounds containing Nitrogen - Online Test
Q1. The following compounds are called
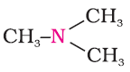

Answer : Option D
Explaination / Solution:
The following compound is tertiary amine.
Q2. The following amine is called as
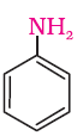

Answer : Option D
Explaination / Solution:
The above compound is aniline and benzeneamine.
Q3. The following amine can be classified as
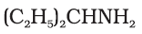
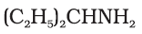
Answer : Option C
Explaination / Solution:
This is primary amine, because the Nitrogen of amine is only connected to one carbon.
Q4. The following amine can be classified as (C2H5)2NH
Answer : Option B
Explaination / Solution:
Nitrogen is attached is two carbons, so this is asecondary amine.
Q5. The following amine can be classified as
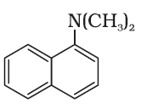

Answer : Option D
Explaination / Solution:
This is tertiary amine but since all the alkyl groups are attachted so it is mixed amine.
Q6. The following reaction takes place in the presence of
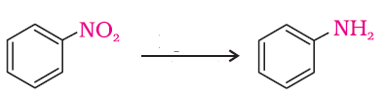

Answer : Option A
Explaination / Solution:
-NO2 group is reduced to –NH2 using H2/Pd.
Q7. Nitro compounds are reduced by iron scrap and hydrochloric acid to yield one of the following compounds.
Answer : Option D
Explaination / Solution:
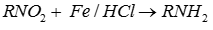
Scrap iron with HCl acts as a reducing agent to reduce nitro group to amine.
Q8. For producing amines, the reaction of nitro compounds with iron scrap is preferred because
Answer : Option C
Explaination / Solution:
This reaction is preferred because on reduction of nitro compouns to amine , Fe will get oxidize to Fe(II) a which on hydrolysis will produce HCl which is required in the reaction.
Q9. Quaternary ammonium salt is formed
Answer : Option A
Explaination / Solution:
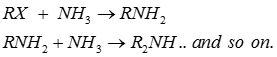
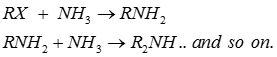
Q10. The main product formed by treating an alkyl or benzyl halide with excess ammonia
Answer : Option B
Explaination / Solution:
The major product is primary amine.
