Chemistry - Online Test
Q1. Which chromium compound is widely used in tanning of leather?
Q2.
Why does H+ ion always get associated with other atoms or molecules?
Answer : Option D
Explaination / Solution:
Loss of an electron from hydrogen atom results in a nucleus of very small size as compared to other atoms or ions.
H+ ion has exceptionally small size, so it can not exist freely.
Q3. Which of the following is correct?
Answer : Option C
Explaination / Solution:
C2H5OH get oxidizes to CH3CHO and –COCH3 group is important for iodoform.
Q4. The kind of isomerism exhibited by [Rh(en)2Cl2][Ir(en)Cl4] and [Ir(en)3][RhCl6] is
Answer : Option D
Explaination / Solution:
There is exchange of ligands between the cationic and anion species in coordination sphere.
Q5. To increase the storage life of butter, the ingredient added is
Answer : Option B
Explaination / Solution:
BHT is added to increase the storage life of butter.
Q6. How much charge is required for the reduction of 1 mol of to Al?
Answer : Option B
Explaination / Solution:
For reduction of 1 mol of to Al , 3 mol of electrons are required so total charge will be 3F.
Q7. The structure of is
Answer : Option C
Explaination / Solution:
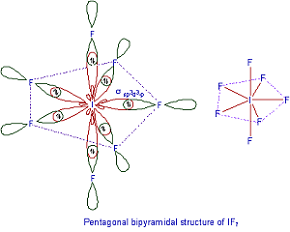
In IF7 out of 7 Flourine atoms 5 of them are placed on a plane in Pentagon shape. In remaining 2 flourines one is placed above the plane and other below the plane each at 90 degrees
Q8. Tetrachloromethane (Carbon tetrachloride)is
Answer : Option B
Explaination / Solution:
CCl4 is degreasing agent.
Q9. Potassium ions are the most abundant cations. They:
Answer : Option C
Explaination / Solution:
Potassium ions are the most abundant cations. They participate in the oxidation of glucose to produce ATP
Q10. In which C-C bond of the inductive effect is expected to be the least?
Answer : Option C
Explaination / Solution:
As distance increases inductive effect also decreases.
