Unit V: Coordination Chemistry - Online Test
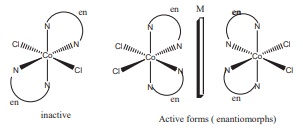
In the complex [M (en)2 (Ox)]
Cl For the central metal ion M3+
The primary valance is = +3
The secondary valance = 6
sum of primary valance and secondary valance = 3+6 = 9
The complex is [M(H2O)5
Cl] Cl2
1000 ml of 1M solution of the
complex gives 2 moles of Cl− ions
1000 ml of 0.01M solution of the
complex will give
[100 ml x 0.01M x 2Cl-] /
[1000 ml x 1M] = 0.002 moles of C− ions
Molecular formula: MSO4Cl.
6H2O .
Formation of white
precipitate with Barium chloride indicates that SO42-
ions are outside the coordination
sphere, and no precipitate with AgNO3 solution indicates that the Cl−
ions are inside the coordination sphere.Since the coordination number of M is
6, Cl− and 5 H2O are ligands, remaining 1 H2O molecular
and SO42- are in
the outer coordination sphere.
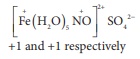
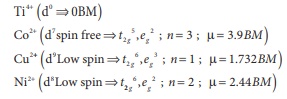
The electronic
configuration t2g3, eg2
[3x(-0.4) + 2(0.6)]Δ0
[-1.2 + 1.2]Δ0 =0