Chemistry - Online Test
Q1.
How many atoms of Oxygen are there in 18g of water?
(Hint: Avagadro’s Number
= 6.02 x atoms/mol)
Answer : Option C
Explaination / Solution:
The problem is based upon the concepts of Avogadro's law
.Accordingly , one mole (ie 18 g ) of water contains
= 6.02 x molecules of water
. Again , since the number of oxygen atoms in one molecule of water ( H2 O )
= 1 atom
the number of oxygen atoms in 6.02 x 1023 molecules of water
.= 6.02 x 1023 atoms
Q2. Name the gas whose formula was established by Sorret.
Answer : Option C
Explaination / Solution:
Sorret established formula of ozone.
Q3. Arrange the following alkyl halides in decreasing order of the rate of elimination reaction with alcoholic KOH
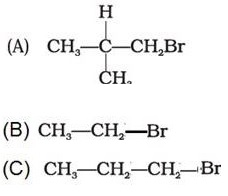

Answer : Option B
Explaination / Solution:
A more substituted alkene is preferred in accordance with the Saytzeff's rule.Hence the order.
Q4. Impurities associated with Bauxite ore is/are
Answer : Option A
Explaination / Solution:
Bauxite ore have silica, TiO2 and Fe2O3 as impurity.
Q5. Calculate the number of kJ of heat necessary to raise the temperature of 60.0 g of aluminium from 35 to 55. Molar heat capacity of Al is 24 J
Answer : Option D
Explaination / Solution:
q=nC ΔT where n= 60.0/27 mole and ΔT = 20 K and C = 24J/mol K
Q6. Packing efficiency for simple cubic structure is
Answer : Option A
Explaination / Solution:
Simple cubic have packing efficiency of 52%.
Q7. One of the following has ns1 as its outermost electronic configuration
Answer : Option D
Explaination / Solution:
The elements of Group 1 (alkali metals) which have ns1 outermost electronic configuration belong to the s-Block Elements.
Q8. Which of the following options is monomer units of the below polymer
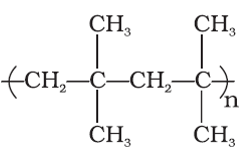

Answer : Option D
Explaination / Solution:
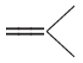
this is monomer unit present in the above given polymer.

this is monomer unit present in the above given polymer.
Q9.
Wave number of yellow radiations having wavelength of 5800 .
Answer : Option B
Explaination / Solution:
Wave number is defined as the reciprocal of wavelength.
where, = wavelength = 5800 Å =
So,
Q10. Sulphur dioxide levels can be reduced by using :
Answer : Option C
Explaination / Solution:
Sulphur dioxide levels can be reduced by using Low sulphur fuels so that less amount of SO2 is produced from them
