Chemistry - Online Test
Q1.
The following compounds are called:
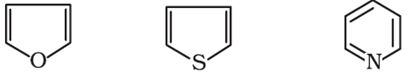

Answer : Option C
Explaination / Solution:
they are aromatic heterocyclic compounds (furan, thiophene and pyridinr respectively).
Q2. Which among the following statement is not true for rate constant of a reaction?
Answer : Option B
Explaination / Solution:
Rate constant is independent of concentration of reactant.
Q3. Pt and Pd adsorb large volume of hydrogen under specific conditions. The hydrogen so adsorbed is called
Answer : Option D
Explaination / Solution:
Adsorption of hydrogen by metal like Pt is occlution and hydrogen is known as occluded hydrogen.
Q4. Density of a gas is found to be 5.46 at 27 at 2 bar pressure. What will be its density at STP?
Answer : Option A
Explaination / Solution:
3g/
Q5. Which of the following amines can be resolved into enantiomers?
Answer : Option B
Explaination / Solution:
2-pentanamine forms enantiomers.
Q6. In alcohols, increase of branching in carbon chain results in boiling point
Answer : Option B
Explaination / Solution:
As the length of carbon chain increases, the surface area of the compound will also increase. Van der Waals dispersion force is proportional to the surface area. So the increase of surface are increases the ability of individual molecules to attract each other. Branching in molecules decreases the surface area thereby decreasing the attractive force between individual molecules. As a result, the boiling point decreases.
Q7. Acidity of can be explained on the basis of which of the following concepts?
Answer : Option D
Explaination / Solution:
Acidity of is explained on the basis of Lewis concept because Boron have vacant p-orbital.
Q8. The use of pressure cooker reduces cooking time because it creates
Answer : Option A
Explaination / Solution:
High pressure increases the boiling point of water so it reduces the cooking time.
Q9. Amines are soluble in
Answer : Option C
Explaination / Solution:
Amines are soluble in water due to hydrogen bonding with water.
Q10. An oxidation number of +1 is found in all their compounds of one of the below given options
Answer : Option A
Explaination / Solution:
Alkali metals have +1 oxidation state.
Because they have only one electron in their outer most shell and by losing that one they can achieve stable configuration, that's why they have +1 oxidation state.
