Chemistry - Online Test
Q1. Acid rain does not cause :
Answer : Option D
Explaination / Solution:
Acid rain does not cause Global warming as it does not release any greenhouse gas in the atmosphere.
Q2. Haemoglobin and chlorophyll contain
Answer : Option D
Explaination / Solution:
Hb contains Fe and Chlorophyll contains Mg.
Q3. Hydrogen resembles halogens in many respects for which several factors are responsible. Of the following factors which one is most important in this respect?
Answer : Option B
Explaination / Solution:
Hydrogen, like the halogens, has one electron short of a complete outer shell and can form H- ions like Cl- and therefore forms ionic compounds with reactive metals – NaH similar in structure to NaCl. Hydrogen can gain one electron to form a duplet.Hydrogen also exists as a diatomic gas like fluorine and chlorine.
Q4. Ketones are reduced to the corresponding alcohols by catalytic hydrogenation to form
Answer : Option B
Explaination / Solution:
Ketones reduction give secondary alcohols.
Q5. Which of the following compound would exhibit coordination isomerism?
Answer : Option D
Explaination / Solution:
Coordination isomerism is exhibited the the compounds in which cationic and anionic species is coordination complex.
Q6. A deodorant is used to reduce body odour due to bacterial decomposition because it is an
Answer : Option C
Explaination / Solution:
A deodorant is intended to inhibit bacterial growth with the use of antiseptic.
Q7. Conductivity of 0.00241 M acetic acid is 7.896 × S . If for acetic acid is 390.5 S , what is its dissociation constant?
Answer : Option D
Explaination / Solution:
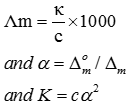
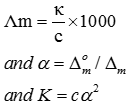
Q8. Which of the following has maximum number of lone pairs associated with Xe?
Answer : Option D
Explaination / Solution:
- n XeF2: Xe has 2 of its electrons in bonding with F. So, since Xe has 8 valence electrons, the no.of lone pairs =
- In XeO3: Xe has 6 of its electrons in bonding with O (bcoz of double bond). So, Xe will have 2 non bonding electrons and therefore 1 lone pair.
- In XeF4: Xe has 4 of its electrons in bonding with F. So, Xe will have 4 non bonding electrons and therefore 2 lone pairs.
- In XeF6: Xe has 6 of its electrons in bonding with F. So, Xe will have 2 non bonding electrons and therefore 1 lone pair.
So XeF2 has the maximum no. of lone pairs associated with Xe.
Q9. p,p’ – Dichlorodiphenyltrichloroethane is a
Answer : Option C
Explaination / Solution:
p,p’ –Dichlorodiphenyltrichloroethane (DDT) is a colorless, tasteless, and almost odorless known for its pesticidal properties and environmental impacts.
Q10. Sodium hydrogencarbonate is known as
Answer : Option A
Explaination / Solution:
is baking soda.
