Chemistry - Online Test
Q1. Identify the type of polymer. —A—A—A—A—A—A—
Answer : Option A
Explaination / Solution:
Since only one monomer unit is present so it is Homopolymer.
Q2. Television pictures result due to
Answer : Option B
Explaination / Solution:
Cathode rays (also called an electron beam or e-beam) are streams of electrons observed in vacuum tubes. If an evacuated glass tube is equipped with two electrodes and a voltage is applied, the glass behind of the positive electrode is observed to glow, due to electrons emitted from and traveling away from the cathode (the electrode connected to the negative terminal of the voltage supply).
Cathode ray tubes (CRTs) use a focused beam of electrons deflected by electric or magnetic fields to create the image in a television set
Q3. Which of these reactions in the atmosphere leads to acid rain ?
Answer : Option B
Explaination / Solution:
2SO2 + O2 + 2H2O ------> 2H2SO4 is the major reaction of acid rain. SO2 emitted from burning of fossil fuels containing S react with O2 and H2O to form the acid leading to acid rain.
Q4. How many carats are in 87.5% gold?
Answer : Option B
Explaination / Solution:
Karat = 24 × (MgMm)Mg= 87.5 and Mm= 100
Q5. In the process of obtaining pure de-mineralised water is passed initially through cation exchange, This makes the water
Answer : Option C
Explaination / Solution:
Cation exchange resin have exchangeable hydrogen ions which makes the water acidic.
Q6. One mole of a symmetrical alkane on ozonolysis gives two moles of an aldehyde having molecular mass of 44u. The alkene is.
Answer : Option A
Explaination / Solution:
2-butene on reductive ozonolysis will give CH3CHO which has molecular mass of 44u.
Q7. Lithiumtetrahydridoaluminate is represented as
Answer : Option A
Explaination / Solution:
Lithiumtetrahydridoaluminate is Li[AlH4]
Q8. A sample of water was found to contain ions. Which of these will be preferred for washing clothes?
Answer : Option D
Explaination / Solution:
Detergents
Q9. The conductivity of 0.20 M solution of KCl at 298 K is 0.0248 S . Calculate its molar conductivity.
Answer : Option C
Explaination / Solution:
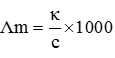
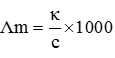
Q10. The maximum number of hydrogen bonds that a molecule of water can have is
Answer : Option B
Explaination / Solution:
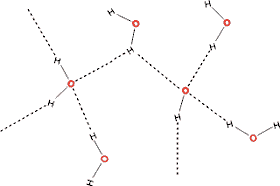
The two hydrogens of the water molecule can form hydrogen bonds with other oxygens in water, and the two lone pair of electrons on oxygen of the water molecule can attract other hydrogens in water. Hence, 4 possible hydrogen bonds.
