Chemistry - Online Test
Q1. Boron is unable to form BF6− ion.
Answer : Option D
Explaination / Solution:
Boron cannot expand its covalency due to absence of d orbitals.
Q2. The common salt or table salt is
Answer : Option C
Explaination / Solution:
Common salt is NaCl.
Q3. The organic compound given below is one of the four options given below. Choose the most appropriate one.
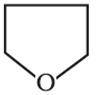

Answer : Option A
Explaination / Solution:
this is aliphatic heterocyclic compound.
Q4. The unit of rate constant for a first order reaction is
Answer : Option C
Explaination / Solution:
Unit of k = mol L−1s−1/ mol L−1
Q5. Which is correct in case of Vander waals adsorption?
Answer : Option B
Explaination / Solution:
Physiadsorption is favoured only at low temperature and high pressure.
Q6.
What will be the pressure exerted by a mixture of 3.2 g of methane and 4.4 g of carbon dioxide contained in a 9 flask at ?
Answer : Option C
Explaination / Solution:
P=P1+P2 (Acc. To Daltons Law) And P1×9=0.2xRT And P2×9=0.1xRT where T= 300K
Q7. The following structure of protein is called
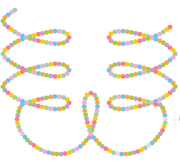

Answer : Option A
Explaination / Solution:
Tertiary structure is this structure.
Q8. One mole of an organic compound 'A' with the formula C3H8O reacts completely with two moles of HI to form X and Y. When 'Y' is boiled with aqueous alkali forms Z. Z answers the iodoform test. The compound 'A' is ______.
Answer : Option D
Explaination / Solution:
Ether react with HI to form alcohol and alkyl iodide. Alcohol on oxidation will give iodoform test.
Q9.
The ionization constant of an acid, , is the measure of strength of an acid. The Ka values of acetic acid, hypochlorous acid and formic acid are respectively. Which of the following orders of pH of 0.1 mol dm-3 solutions of these acids is correct?
Answer : Option B
Explaination / Solution:
pH=-log(cα) , Ka= . Lesser is the pH value greater is acidity. Also greater is Ka, lesser will be acidity.
Q10. Which among the following is soluble in water?
Answer : Option C
Explaination / Solution:
Because of hydrogen bonding.
