Chemistry - Online Test
Q1. Greater the value of KH, lower will be the
Answer : Option C
Explaination / Solution:
Pgas= KH× Xgas.Xgas
is measure of solubility of gas.
is measure of solubility of gas.
Q2. Which of the following amines can be resolved into enantiomers?
Answer : Option D
Explaination / Solution:
2-pentamine will have a chiral center.
Q3.
In the free or the uncombined state, each atom in O2O3,P4,S8 and Mg has the oxidation number
Answer : Option D
Explaination / Solution:
In free or uncombined state each element has zero oxidation state.
Q4. The molar mass of Al2O3 is
Answer : Option A
Explaination / Solution:
Molar mass
= [ 2 x ( atomic mass of Al ) , 3 x ( atomic mass of O ) ]u
=
= ( 54 + 48 ) u
= 102 u.
Q5. Which of the following is prepared by cyanamide process?
Answer : Option D
Explaination / Solution:
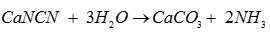
Q6. Which of the following will not show geometrical isomerism?
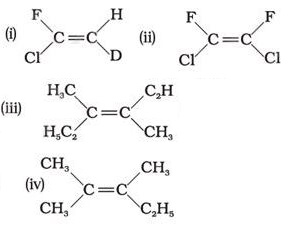

Answer : Option C
Explaination / Solution:
This is because geometrical isomerism is not possible if three groups are same.
Q7. In the froth floatation process for the concentration of ore, the particles float because
Answer : Option B
Explaination / Solution:
In froth floatation ore particles get wetted with oil and comes to the surface with froath.
Q8. A reaction, A + B → C + D + q is found to have a positive entropy change. The reaction will be
Answer : Option C
Explaination / Solution:
ΔG = ΔH -T ΔS
ΔS is positive and ΔH is negative as heat is liberated in the reaction.
so ΔG is negative hence reaction will be spontaneous at all temperature.
Q9. Volume of unit cell occupied in face centered cubic arrangement is
Answer : Option D
Explaination / Solution:
Packing Fraction is 74%
Q10. The outer electronic configuration of f - Block elements are:
Answer : Option C
Explaination / Solution:
The two rows of elements at the bottom of the Periodic Table, called the Lanthanoids, Ce(Z = 58) – Lu(Z = 71) and Actinoids, Th(Z = 90) – Lr (Z = 103) are characterised by the outer electronic configuration (n-2)f1-14 (n-1)d0-1ns2. The last electron added to each element is filled in f- orbital.
