Chemistry - Online Test
Q1. Octet of electrons, represents a particularly stable electronic arrangement. Atoms achieve the stable octet when they are linked by chemical bonds. This rule is associated with one of the following theories
Answer : Option D
Explaination / Solution:
G.N.Lewis, proposed the octet rule to explain the valence electron sharing between atoms that resulted in a bonding type with the atoms attaining noble gas electronic configuration.
The statement is : "a bond is formed between two atoms by mutual sharing of pairs of electrons to attain a stable outer-octet of electrons for each atom involved in bonding". This type of valence electron sharing between atoms is termed as covalent bonding.
Q2. Necessary conditions for any system to be aromatic:
Answer : Option D
Explaination / Solution:
Aromaticity condition involves huckel rule (4n+2) Pie electrons. And proper delocalization of pie electron.
Q3. Washing soda is
Answer : Option C
Explaination / Solution:
Washing soda is
Q4.
represents methoxypropane and ethoxyethane
Answer : Option D
Explaination / Solution:
rearrangement of carbon atoms around the functional group is known as metamerism.
Q5.
The reactionobeys the rate equationwhat would be the order of this reaction?
Answer : Option B
Explaination / Solution:
Order of reaction with respect to a is x and wrt to b is y so total order of reaction is x+y.
Q6. Adsorption is a
Answer : Option A
Explaination / Solution:
Adsorption is entirely a bulk phenomenon while absorption is bulk phenomenon.
Q7.
The drain cleaner, Drainex contains small bits of aluminum which react with caustic soda to produce dihydrogen. What volume of dihydrogen at and one bar will be released when 0.15g of aluminum reacts?
Answer : Option B
Explaination / Solution:
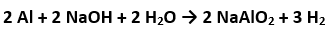
Moles of Al= 0.15/27 =0.0055 moles And 2 mole of Al produces 3 moles of so 0.0055 moles will produce 3/2x0.0055 moles of =0.00825 moles. Now PV=nRT where n=0.00825 , P=1 bar and T=293K.
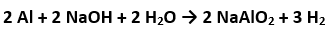
Moles of Al= 0.15/27 =0.0055 moles And 2 mole of Al produces 3 moles of so 0.0055 moles will produce 3/2x0.0055 moles of =0.00825 moles. Now PV=nRT where n=0.00825 , P=1 bar and T=293K.
Q8. The biological polymer is
Answer : Option A
Explaination / Solution:
Nucleic acid is biological polymers.
Q9. An organic compound X is oxidised by using acidified K2Cr2O7. The product obtained reacts with Phenyl hydrazine but does not answer silver mirror test. The possible structure of X is
Answer : Option C
Explaination / Solution:
Secondary alcohol on oxidation forms ketone which reacts with hydrazine bus doesnot gives silver mirror test.
Q10. The pH of neutral water at is 7.0. As the temperature increases, ionisation of water increases, however, the concentration of ions and ions are equal. What will be the pH of pure water at
Answer : Option C
Explaination / Solution:
pH of water will be around 7 only. Water will be neutral even at 60°C
