Chemistry - Online Test
Towards the end of the 19th century Joseph J.Thomson (1856-1940) was studying electric discharges at the well-known Cavendish laboratory in Cambridge, England. Several people had been studying the intriguing effects in electric discharge tubes before him. Spectacular glows could be observed when a high voltage was applied in a gas volume at low pressure. It was known that the discharge and the glow in the gas were due to something coming from the cathode, the negative pole of the applied high voltage. Thomson made a series of experiments to study the properties of the rays coming from the cathode. He observed that the cathode rays were deflected by both electric and magnetic fields - they were obviously electrically charged. By carefully measuring how the cathode rays were deflected by electric and magnetic fields, Thomson was able to determine the ratio between the electric charge (e) and the mass (m) of the rays. Thomson's result was
The particle that J.J.Thomson discovered in 1897, the electron, is a constituent of all the matter we are surrounded by. All atoms are made of a nucleus and electrons. He received the Nobel Prize in 1906 for the discovery of the electron, the first elementary particle.
Pollutant gases dissolve in water in atmosphere to form Acid rain.
2SO2 + 2H2O + O2 -------> 2H2SO4
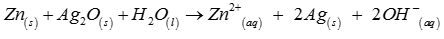
Determine E^ofor the reaction.