Chemistry - Online Test
Q1. of combustion of methane is - X kJ . The value of is
Answer : Option A
Explaination / Solution:
CH4 (g) + 2O2 (g) → CO2 (g) + 2H2O (l)
We know, RT
Δng is negative as product water in combustion reaction is in liquid state.
This implies ΔHo will be more negative than ΔUo
Hence, ΔHo < ΔUo
Q2. To which block of elements in the periodic table belongs?
Answer : Option D
Explaination / Solution:
d-block element have the general outermost electronic configuration as (n-1)d1-10 ns0-2.
Q3. Which of the following monomers form biodegradable polymers?
Answer : Option D
Explaination / Solution:
Glycine + amino caproic acid= nylon 2-nylon 6 3 – hydroxybutanoic acid + 3 – hydroxypentanoic acid = PHBV They are biodegradable.
Q4. One of the following scientists was the first to discover electron
Answer : Option B
Explaination / Solution:
Electron was discovered by J. J. Thomson in 1897 when he was studying the properties of cathode ray. J. J. Thomson won Nobel Prize in 1906 for discovering the elementary particle electron.
Q5. Which of these is not released from burning fossil fuels?
Answer : Option C
Explaination / Solution:
Copper oxide can never be released during burning of fossil fuels because they do not contain copper in it which would be released as copper oxide after combustion.
Q6. Which of the following methods is based on distribution law?
Answer : Option B
Explaination / Solution:
Parkes process is a process for removing silver from Pb. It is an example of Liquid liquid extraction.s
Q7. H2O2 as an antispectic is used in the name of
Answer : Option B
Explaination / Solution:
Perhydrol is a solution of hydrogen peroxide (H2O2) at a concentration of 30% of weight by volume(w/v) or 100 volume strength.
Perhydrol is a mild antiseptic used on the skin to prevent infection of minor cuts, scrapes, and burns. It may also be used as a mouth rinse to help remove mucus or to relieve minor mouth irritation (e.g., due to canker/ cold sores, gingivitis). This product works by releasing oxygen when it is applied to the affected area. The release of oxygen causes foaming, which helps to remove dead skin and clean the area.
Q8.
Identify the product in the reaction
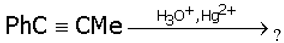
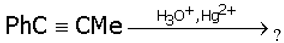
Answer : Option A
Explaination / Solution:
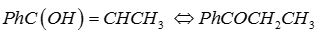
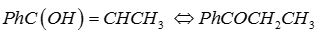
Q9. Nutritive value is imparted to the food by
Answer : Option C
Explaination / Solution:
None of above impart nutritive value to food.
Q10. Which of the following species is expected to be colourless?
Answer : Option C
Explaination / Solution:
Ti is in d0 state so no d - d transition.
