Chemistry - Online Test
Q1. Which of the following statements is correct?
Answer : Option A
Explaination / Solution:
Ice and water kept in a perfectly insulated thermos flask (no exchange of heat between its contents and the surroundings) at 273K and the atmospheric pressure are in equilibrium state and the system shows interesting characteristic features. We observe that the mass of ice and water do not change with time and the temperature remains constant.
Q2. Arrange the following compounds in increasing order of their acid strength: Propan – 1 – ol, 2,4,6 – trinitrophenol, 3 – nitrophenol, 3,5 – dinitrophenol,phenol, 4 – methylphenol.
Answer : Option D
Explaination / Solution:
Phenols are more acidic than alcohols Also in phenols if –M groups like –NO2 are arranged then acidity increases. And –M operates only at o/p position. At m only –I operates.
Q3. Mathematical expression relating molarity and molality is
Answer : Option A
Explaination / Solution:
m= moles of solute / mass of solvent. M= moles of solute / volume of solution.
Q4. The following compound is called
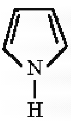

Answer : Option D
Explaination / Solution:
This is pyrrole, a heterocyclic compound.
Q5. Electrochemical processes for extraction of highly reactive metals and non-metals involves one of the following. Choose the most appropriate option.
Answer : Option D
Explaination / Solution:
It involves redox process.
Q6.
The gram molar mass of is
Answer : Option A
Explaination / Solution:
The gram molar mass of Ca CO3 is calculated by ,
(i) adding up the atomic masses of Ca , C & 3 O atoms & ,
(ii) representing the molar mass in grams.
Thus , gram molar mass of Ca C O3
= [ atomic mass of Ca , atomic mass of C , 3 x atomic mass of O ]
=
= ( 40 + 12 + 48 ) g
=100g
It should be noted that ,
atomic mass of Ca
= 12
atomic mass of C
= 12
atomic mass of O
= 16
Q7. Hydrogen sulphide reacts with lead acetate forming a black compound which reacts with hydrogen peroxide to form another compound. The colour of the compound is
Answer : Option C
Explaination / Solution:
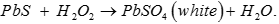
Q8. Ionization enthalpy is lowest in
Answer : Option B
Explaination / Solution:
Metals have low ionization energy because of there tendency to lose electrons.
Q9. The increasing order of reduction of alkyl halides with zinc and dilute HCl is
Answer : Option C
Explaination / Solution:
As we move from I to Br to Cl,atomic size decreases,it becomes difficult to release the halogen as the bond length decreases.Hence I- is the best leaving group among halogens and reacts the fastest.
Q10. Cinnabar is an ore of
Answer : Option B
Explaination / Solution:
HgS is cinnabar.
