Chemistry - Online Test
Q1. Emf of the cell
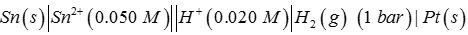
at 298 K is
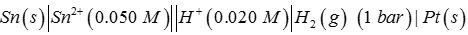
at 298 K is
Answer : Option B
Explaination / Solution:
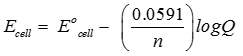
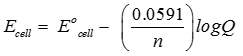
Q2. The criterion of overlap of atomic orbitals is the main factor for the formation of
Answer : Option D
Explaination / Solution:
The valence bond theory is introduced to describe bonding in organic molecules. In this model, bonds are considered to form from the overlapping of two atomic orbitals on different atoms, each orbital containing a single electron.
In general, greater the overlap, stronger is the bond formed between the two atoms. Thus, according to the orbital overlap concept, atoms combine together by overlapping their orbital and thus forming a lower energy state where their valence electrons with opposite spin, pair up to form covalent bond.
Q3. For alkanes containing odd number of carbon atoms, Wurtz reaction is not preferred. Because
Answer : Option C
Explaination / Solution:
In wurtz reaction many side products are formens whwn two different alkyl halides are used.
Q4. In the preparation of Sodium carbonate the by- product formed is:
Answer : Option D
Explaination / Solution:
is the by product.
Q5. The principle involved in paper chromatography is
Answer : Option D
Explaination / Solution:
Paper chromatography is based on the principle of partition.
Q6. Rate of reaction does not remain constant throughout because
Answer : Option C
Explaination / Solution:
Rate of reaction is dependent on concentration of reactant
Q7. Which catalyst is used in Haber’s process?
Q8. Pressure of 1 g of an ideal gas A at 27 ∘C is found to be 2 bar. When 2 g of another ideal gas B is introduced in the same flask at same temperature the pressure becomes 3 bar. Relationship between their molecular masses is
Answer : Option C
Explaination / Solution:
PV=(m/M)RT
Q9. The enzyme which dissolved blook clot is.
Answer : Option C
Explaination / Solution:
Streptokinase enzyme help in dissolving blood clot.
Q10. When hydrochloric acid is added to cobalt nitrate solution at room temperature, the following reaction takes place and the reaction mixture becomes blue. On cooling the mixture it becomes pink. On the basis of this information mark the correct answer.
(l )
(l )
Answer : Option C
Explaination / Solution:
This reaction is endothermic.
