Chemistry - Online Test
Q1. Cassiterite is the chief ore of
Answer : Option D
Explaination / Solution:
SnO2 is formula of casserite.
Q2. The enthalpies of all elements in their standard states are:
Answer : Option C
Explaination / Solution:
The enthalpies of formation of all elements in their standard states is taken as zero.
Q3. The outer electronic configuration of a p- block element is:
Answer : Option C
Explaination / Solution:
In the p-block elements, the last electron enters in p-orbital of valence shell and electronic configuration of valence shell is ns2 np1-6.
Q4. The correct name of the polymer below is:
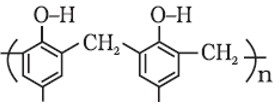

Answer : Option B
Explaination / Solution:
This is Bakelite.
Q5. Cathode rays are discharged under one of the following conditions:
Answer : Option C
Explaination / Solution:
Production of cathode rays Cathode rays are produced in the discharge tube by applying the following conditions:
- A high potential difference (>1200 V) is applied across the two aluminium electrodes.
- The length of the tube is 30 cm and the diameter is 3 cm.
- Pressure inside the tube is maintained below 0.01 mm of Hg.
Q6. The Earth does not directly provide which of man’s basic needs :
Answer : Option A
Explaination / Solution:
The Earth does not directly provide Shelter which is man's basic need. we have to use different resources on earth available in different forms to convert them into objects that can be used as a shelter
Q7. Red hot steel rods on suddenly immersing in water become
Answer : Option B
Explaination / Solution:
Red hot steel rods on suddenly immersing in water become hard and brittle.
Q8. Calgon is used in
Answer : Option D
Explaination / Solution:
Calgon is a process of removing permanant hardness of water. Calgon is a complex salt, Sodium hexametaphospahte Na(PO3)6 or Na6P6O18 .It acts as a ion exchange resin. It is used for softening of water.When hard water containing Ca and Mg ions is reacted with Calgon, it displaces these ions with Sodium ion, by forming a complex with itself and thus the water becomes free of these ions which cause hardness.
Q9. Acetone is treated with excess of ethanol in the presence of hydrochloric acid. The product obtained is:
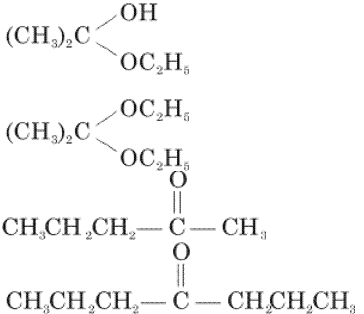

Answer : Option D
Explaination / Solution:
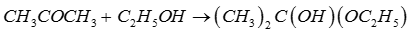
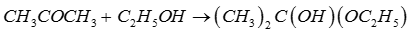
Q10. A patient was suffering from severe pain after a major operation. To reduce his pain, the doctor administered
Answer : Option C
Explaination / Solution:
Narcotic analgesic are given to reduce the pain.
