Chemistry - Online Test
The reaction is not redox because there is no oxidation or reduction of any element (i.e. no change in oxidation state/number).
Ba in BaCl2 is at a +2 charge because it is in group 2. On the other side of the equation Ba in BaSO4 is still +2. Cl2 on the first side of the equation is -2 (as 2 chlorines) and Cl is -2 on the other side as there are 2 of them.
H on both sides are both +1 (but technically +2 as there are two of them).
SO4 does not change oxidation number and always remains -2.
So, the reaction is not a redox reaction.
------ +----- → ------ Ba + ------ .
The above equation can be balanced by inserting the following in blank spaces from LHS to RHS respectively.
The given equation gets stoichiometrically balanced when the coefficients ( 1 , 6 , 3 , 2 ) starting from LHS to RHS are inserted in given blank spaces .
Thus , the balanced equation for the reaction is ,
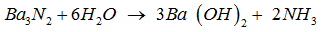
Arrange the halogens in order of their increasing reactivity with alkanes.
