Chemistry - Online Test
Q1. Drug inhibits the enzymatic action by attaching itself to allosteric site, which is
Answer : Option A
Explaination / Solution:
Allosteric site is site on enzyme apart from active site where inhibitors attach.
Q2. Which is used in cancer therapy?
Answer : Option A
Explaination / Solution:
Cis Platin ( cis – [Pt(NH3)2(Cl)2] ) is a anti cancer drug.
Q3.
Emf of the cell
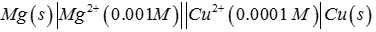
at 298 K is
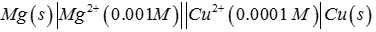
at 298 K is
Answer : Option D
Explaination / Solution:
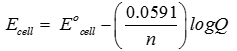
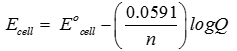
Q4. Ionic crystalline compounds formed by ion-formation by electron transfer proves
Answer : Option D
Explaination / Solution:
Ionic crystalline compounds formed by ion-formation by electron transfer proves electronic theory of chemical bonding.
Q5. Isomers of the compoundC4H9Br are
Answer : Option A
Explaination / Solution:
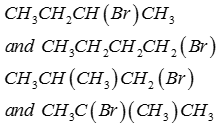
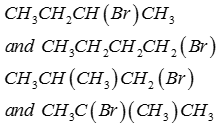
Q6. Why is paramagnetic ?
Answer : Option C
Explaination / Solution:
is a superoxide ( 3 electron species) so has one unpaired electron thus it is paramagnetic.
Q7. The fragrance of flowers is due to the presence of some steam volatile organic compounds called essential oils. These are generally insoluble in water at room temperature but are miscible with water vapour in vapour phase. A suitable method for the extraction of these oils from the flowers is:
Answer : Option C
Explaination / Solution:
A suitable method for the extraction of these oils from the flowers is Steam distillation
Q8. The metabolism of hormones in human body is an example of
Answer : Option C
Explaination / Solution:
It is pseudo first order reaction.
Q9. When 50 mL of 1M oxalic acid is mixed with 0.5 g of wood charcoal, the final concentration of the solution after adsorption is 0.5 M. The amount of the oxalic acid adsorbed per gram of charcoal is
Answer : Option A
Explaination / Solution:
Amount of oxalic acid adsorbed = (0.5x50)/1000 Mass of oxalic acid adsorbed per gram of charcoal = (0.0025x126)/0.5
Q10. What will be the minimum pressure required to compress 500 of air at 1 bar to 200 at
Answer : Option A
Explaination / Solution:
Using ideal gas equation, PV=nRT,
since T is constant, we have
