Chemistry - Online Test
Q1. Arrange the following in decreasing order of their boiling points. (A) n–butane (B) 2–methylbutane (C) n-pentane (D) 2,2–dimethylpropane
Answer : Option C
Explaination / Solution:
Boiling point depends on molecular mass and surface area.As the number of carbon atoms increase,boiling point increases ,hence n-butane has the minimum boiling point.Among isomeric alkanes,boiling point decreases with branching.Hence the order.
Q2. Which metal ores occur in lakes and sea?
Answer : Option A
Explaination / Solution:
Halides ores occur in lakes and sea.
Q3. For an adiabatic change
Answer : Option B
Explaination / Solution:
An adiabatic process would not allow exchange of heat between the system and surroundings, hence q=0.
Q4. Generally, the first ionization energy increases along a period including some exceptions. One which is NOT an exception is ______.
Answer : Option B
Explaination / Solution:
The high ionization energy of B, N and Mg is due to the presence of stable electronic configuration in these atoms as compared to the next element in the period. For ex. Be (Is2 2s2) has competely filled orbitals as compared to B (1s2 2s2 2p1) where there is one electron in p-orbital that can easily be lost. Therefore, B has a lower ionization energy than Be.
Q5. Hydrocarbons are prepared from Grignard reagent by
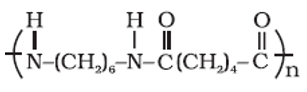
Answer : Option B
Explaination / Solution:
This is nylon 6,6 formed by polymerization of adipic acid and hexamethylenediamine.
Q6. How many neutrons and protons are there in the nucleus?
Answer : Option A
Explaination / Solution:
Number of protons = Atomic number =6
Number of neutrons = atomic mass - no of protons = 13-6 = 7
Q7. Three elements needed for the healthy growth of plants are
Answer : Option D
Explaination / Solution:
Nitrogen , phosphorus, and potassium are needed.
Q8. Mohr’s salt is a better volumetric agent then ferrous sulphate because
Answer : Option B
Explaination / Solution:
Mohr’s salt is a better volumetric agent then ferrous sulphate because
it is less readily oxidised in the solid state. Also Mohr salt is more
stable than FeSO4 .
Q9. is an example of
Answer : Option B
Explaination / Solution:
Water present in the interstitial sites in a crystal lattice is known as interstitial water. They are not held by hydrogen bonding with the other elements. If they are bonded they are called H-bonded hydrates.
Barium chloride is a white crystalline powder. It contains two molecules of water. It is one of the most important water soluble salts of Barium.
Q10. Benzaldehyde and acetone can be best distinguished using
Answer : Option A
Explaination / Solution:
Aldehydes show tollen’s test while acetone which is a ketone doesnot give tollen’s test.
