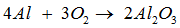Chemistry - Online Test
Lucas Test is a test which is used to distinguish between primary, secondary and tertiary alcohols. This test is carried out with the help of Lucas reagent, which is a solution of anhydrous Zinc Chloride and concentrated hydrochloric acid (ZnCl2 + HCl). It is based on the difference between the reactivity of primary, secondary and tertiary alcohols with hydrogen halides.
If an excess of is treated with , then_____will be produced wherein the oxidation number of P is +3. The compound is
Whenever a reaction between an oxidising agent and a reducing agent is carried out, a compound of lower oxidation state is formed if the reducing agent is in excess and a compound of higher oxidation state is formed if the oxidising agent is in excess. This can be illustrated as follows:
P4 and F2 are reducing and oxidising agents respectively. If an excess of P4 is treated with F2, then PF3 will be produced, wherein the oxidation number (O.N.) of P is +3.
Consider the unbalanced chemical equation ,
----- Al + ------ → ------ .
The equation for above reaction can be balanced by inserting the following in blank spaces