Chemistry - Online Test
Q1. The compound obtained when acetaldehyde reacts with dilute aqueous sodium hydroxide exhibits:
Answer : Option C
Explaination / Solution:
CH3 CH=CHCHO is the product which is suitably substituted so will show geometrical isomer.
Q2. The water free from ions
Answer : Option D
Explaination / Solution:
Water free from and ions is soft water.
Q3. The correct IUPAC name of Mn3(CO)12 is
Answer : Option A
Explaination / Solution:
Ligands are named 1st followed by the name of central metal ion.
Q4. Calculate the standard cell potentials of galvanic cell, ∆_r G^oand equilibrium constant of the reactions if the reaction is
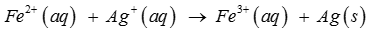
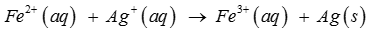
Answer : Option A
Explaination / Solution:
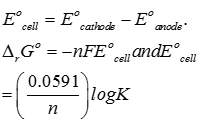
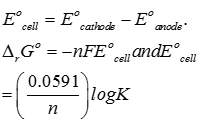
Q5. Polarity in a molecule and hence the dipole moment depends primarily on electronegativity of the constituent atoms and shape of a molecule. Which of the following has the highest dipole moment?
Answer : Option C
Explaination / Solution:
CO2 is linear, so even though the C−O bonds have individual dipole moments, the overall dipole moment is zero as these cancel out (they point in opposite directions, as shown in the diagram below).
On the other hand, H2O is "bent", which means that the individual dipole moments of the bond are at an angle to each other and because of Presence of lp on head of O atom in H2O therefore it has max dipole moment.
Q6. Name the following aryl halide as per the IUPAC system
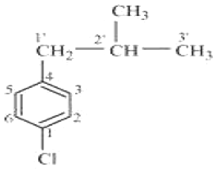

Answer : Option D
Explaination / Solution:
At position 1 chloro group is present and at 2-methylpropyl is present.
Q7. All alkali metal halides are soluble in water except
Answer : Option A
Explaination / Solution:
Greater the lattice energy, higher is the melting point of the MX (metal halide) and lower is its solubility
Q8. In which of the following, functional group isomerism is not possible?
Answer : Option B
Explaination / Solution:
alkyl halides donot give alkyl halides.
Q9. Rate of ionic reactions are generally
Answer : Option D
Explaination / Solution:
Ionic reactions occur very fast.
Q10. Ammonia is adsorbed on activated charcoal to a larger extent as compare to CO because
Answer : Option C
Explaination / Solution:
Ammonia is easily liquefiable because of h bonding interactions.
