Chemistry - Online Test
Q1. According to Faraday’s Second Law of Electrolysis the amounts of different substances liberated by the same quantity of electricity passing through the electrolytic solution are proportional
Answer : Option B
Explaination / Solution:
The amounts of different substances liberated by the same quantity of electricity passing through the electrolytic solution are proportional to their chemical equivalent weights.
Q2. Structures of . ion given below explains the phenomenon of _____________.
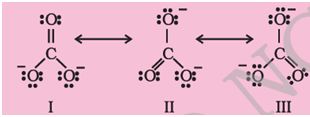

Answer : Option D
Explaination / Solution:
Resonance is the phenomenon when a molecule can be represented by 1 or more structure differing in the position of bonds and charge.
Q3. The following compound is:
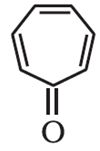

Answer : Option D
Explaination / Solution:
Tropone or 2,4,6-cycloheptatrien-1-one is a non-benzenoid aromatic compound. The compound consists of a ring of seven carbon atoms with three conjugated alkene groups and a ketone group.
Q4. Carbon – halogen bond of alkyl halides is responsible for their nucleophilic substitution, elimination and their reaction with metal atoms to form organometallic compounds because of their
Answer : Option D
Explaination / Solution:
R—X is polar.
Q5. Flame photometry is used to determine the concentration of the
Answer : Option D
Explaination / Solution:
Flame photometry is used to determine the concentration of alkali metalions, alkaline earth metals are quite sensitive to flame photometry.
Q6. The expression which relates the rate of reaction to the concentration of the reactants are called
Answer : Option B
Explaination / Solution:
Rate law states that rate of reaction is dependent on concentration of reactant raise to power order of reaction.
Q7. Fog is a colloidal solution of
Answer : Option A
Explaination / Solution:
Dispersed phase is liquid, dispersion medium is gas.
Q8. In the kinetic molecular theory attraction between molecules of the gas is
Answer : Option D
Explaination / Solution:
The Kinetic Theory of Gases assumes that no intermolecular forces exerts between the gas molecules.
Q9. the concentrations in an equilibrium mixture are related by the following equilibrium equation, Where Kc is called as
Answer : Option B
Explaination / Solution:


Q10. Glycogen is formed in
Answer : Option C
Explaination / Solution:
Glycogen is how energy is stored in animals
