Chemistry - Online Test
The Cannizzaro reaction, named after its discoverer Stanislao Cannizzaro, is a chemical reaction that involves the base-induced disproportionation of an aldehyde lacking a hydrogen atom in the alpha position.
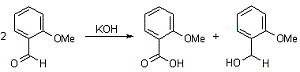
This redox disproportionation of non-enolizable aldehydes to carboxylic acids and alcohols is conducted in concentrated base.

A common characteristic of most Alkali metal is their ability to displace H2 (g) from water. This is represented by their large, negative electrode potentials. In this event, the Group 1 metal is oxidized to its metal ion and water is reduced to form hydrogen gas and hydroxide ions. The general reaction of an alkali metal (M) with H2O (l) is given in the following equation:
2M(s)+2H2O(l)⟶2M+(aq)+2OH−(aq)+H2(g)
From this reaction it is apparent that OH- is produced, creating a basic or alkaline environment. Group 1 elements are called alkali metals because of their ability to displace H2 (g) from water and create a basic solution.Alkali metals are also known to react violently and explosively with water. This is because enough heat is given off during the exothermic reaction to ignite the H2(g)
