Chemistry - Online Test
The effect of Heisenberg uncertainty principle is significant only for motion of microscopic objects, it is negligible in case of macroscopic objects. For example, we apply the concept of uncertainty to an object of mass 1 milligram.
The value of is very small and insignificant in this case. Therefore, when we deal with heavier objects the associated uncertainties have no real consequences and can be neglected.
Avagadro's constant represents the number of entities ( viz, atoms , molecules or ions ) present in 1 mole of a substance .
The constant ( denoted by N )
=6.022 x 1023
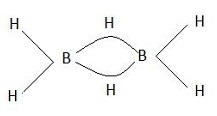
Diborane is one of the following
Diborane is said to be electron deficient because boron require 5 electrons to complete its octet. But in this molecule, each boron atom is bonded to 2 terminal hydrogen atoms each. Also, the two boron atoms are held together by two hydrogen atoms as shown below: