Chemistry - Online Test
Q1. Honey belongs to one of the following category
Answer : Option D
Explaination / Solution:
Honey is a carbohydrate
Q2. benzaldehye differs from acetaldehyde in that
Answer : Option C
Explaination / Solution:
Fehling's Solution Test: When acetaldehyde is heated with Fehling solution, red ppt. of cuprous oxide are formed. But benzaldehyde does not give red ppt. with fehling solution.
Reaction are
CH3CHO + 2Cu2+ + 5OH-
CH3COO- + Cu2O + 3H2O
C6H5CHO + 2Cu2+ + 5OH-
Q3. The boiling point of isomeric branched chain alkene is
Answer : Option C
Explaination / Solution:
As surface area decreases with branching,the boiling point also decreases.
Q4.
Arrange the following in decreasing order of their basic strength :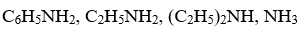
Answer : Option B
Explaination / Solution:
Alkyl amines and ammonia are more basic than aniline because in aniline lone pair on N is in resonace with benzene ring so is less available.
Also more 20 alkyl amines are more basic than primary amine and ammonia due to +I effect of the alkyl group which increases basicity.
Q5. Solubility of gas decreases with
Answer : Option C
Explaination / Solution:
Because of increase in kinetic energy of gas molecules with temperature there tendency to escape from the liquid will increase.
Q6. A metal in a compound can be displaced by another metal in the uncombined state. Which metal is a better reducing agent in such a case?
Answer : Option B
Explaination / Solution:
Reducing agent is one in which element undergo oxidation itself and make the other substance reduce.
Q7. Substances that are strongly attracted by applied magnetic field and can be permanently magnetized are
Answer : Option D
Explaination / Solution:
Ferromagnetic substance get permanently magnetised in applied Magnetic Field.
Q8. Addition polymers formed by the polymerisation of a single monomeric species
Answer : Option C
Explaination / Solution:
When single monomer is present polymer is homopolymer.
Q9.
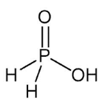 is the structure of
is the structure of
 is the structure of
is the structure of
Answer : Option C
Explaination / Solution:
H3PO2 is Hypophosphorus acid. It has one P-OH, two P-H and one P=O bonds.
Q10. According to Hess’s Law if a reaction takes place in several steps then its standard reaction enthalpy is
Answer : Option C
Explaination / Solution:
Hess’s Law states that the sum of the standard enthalpies of the intermediate reactions into which the overall reaction may be divided at the same temperature.
