Chemistry - Online Test
Q1. Dettol is a mixture of
Answer : Option B
Explaination / Solution:
Dettol is mixture of chloroxylenol and terpineol.
Q2. In which of the following compounds, the metal is in the lowest oxidation state?
Answer : Option A
Explaination / Solution:
CO is neutral so Mn has zero oxidation state.
Q3. According to Faraday’s First Law of ElectrolysisThe amount of chemical reaction which occurs at any electrode during electrolysis by a current is proportional to the
Answer : Option B
Explaination / Solution:
Faraday’s first law of electrolysis states that amount of substance deposited at any of the electrode is directly proportional to amount of charge passed.
Q4. , CO and are isoelectronic molecules. Their respective bond order is :
Answer : Option D
Explaination / Solution:
Total number of electrons in N2 molecule is 7+ 7= 14.
As per the formula Bonded pair of electrons Nb:
Total 10 electrons.
Anti bond pairs of electrons Na: Total 4 electrons.
Bond Order (B.O.) =
Similarly bond order of CO And NO+ is 3
Q5. Benzene and other related ring compounds (benzenoid) such as given below are called:
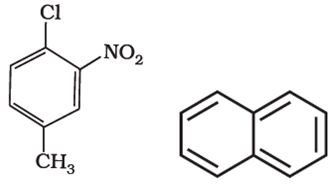

Answer : Option C
Explaination / Solution:
they are aromatic system as they have benzene ring intact.
Q6. Upon oxidation, one of the following halogen containing compounds forms an extremely poisonous gas – – + O2 → 2 COCl2 + 2HCl
Answer : Option C
Explaination / Solution:
CHCl3 on reaction with oxygen forms phosgene. Chloroform
decomposes when it exposed to sunlight.it decomposes to a harmful gas
phosgene(COCl2) Chloroform decomposes when it exposed to sunlight.it decomposes to a harmful gas phosgene(COCl2)
Q7.
The rate of chemical reaction becomes double for every 10o rise in temperature because of
Answer : Option A
Explaination / Solution:
With increase in temperature molecular collisions increases therefore rate of reaction doubles.
Q8. Enzymes are
Answer : Option A
Explaination / Solution:
Enzymes are biological catalyst, they catalize different chemical reactions in body.
Q9. Dalton’s Law of Partial Pressures states that the total pressure exerted by the mixture of non-reactive gases is equal to the
Answer : Option D
Explaination / Solution:
The question is based on the statement of Dalton Law of Partial pressures for Gases which states that total pressure exerted by mixture of non reacting gases is equal to the sum of partialpressure of individual gases.
Q10. For dissolution of gases in liquids, the concentration of a gas in liquid is.
Answer : Option C
Explaination / Solution:
For dissolution of gases in liquids, the concentration of a gas in liquid is proportional to the pressure of the gas over the liquid
