Chemistry - Online Test
Q1. Which of the following is the correct ideal gas law
Answer : Option D
Explaination / Solution:
PV=nRT is the correct Ideal Gas equation.
Here, P : Pressure
V : Volume
n : Number of Moles
R : Universal Gas Constant
T : Temperature
Q2. The aqueous solution of sugar does not conduct electricity. However, when sodium chloride is added to water, it conducts electricity. How will you explain this statement on the basis of ionisation and how is it affected by concentration of sodium chloride?
Answer : Option B
Explaination / Solution:
Sugar being a non-electroyte, does not ionize in water, whereas NaCl ionizes completely in water and produce Na+ and Cl- ions, which help in the conduction of electricity and when concentration of NaCl is increased , more Na+ and Cl- ions will be produced, hence conductance increases.
Q3. The cyclic form of fructose is called
Answer : Option A
Explaination / Solution:
Cyclic form of fructose is called furanose.
Q4. In Q 14, the catalyst used is not one of the following
Answer : Option C
Explaination / Solution:
Ag cannot act as catalyst.
Q5. p – toluenesulphonyl chloride is used to
Answer : Option C
Explaination / Solution:
p – toluenesulphonyl chloride can also be used to distinguish between , and amines in replacement of benzenesulphonylchloride.
Q6. Stereoisomeric alkenes due to different arrangement of atoms or groups in space are referred to as
Answer : Option A
Explaination / Solution:
Isomers which have the same structural formulae but difffer in the relative spatial arrangements of atoms or groups around the double bond are called geometric isomers.athus geometrical isomers are atype of stereoisomers.
Q7. The sum of all mole fraction for a mixture is always equal to
Answer : Option A
Explaination / Solution:
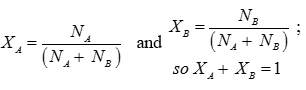
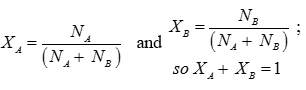
Q8. All combustion reactions, which make use of elemental dioxygen, as well as other reactions involving elements other than dioxygen are:
Answer : Option B
Explaination / Solution:
A combination reaction (also known as a synthesis reaction) is a reaction where two or more elements or compounds (reactants) combine to form a single compound (product). Such reactions may be represented by equations of the following form: X + Y → XY.
Q9.
The general electronic configuration of ns2 exit in:
Answer : Option A
Explaination / Solution:
The general electronic configuration of ns2 exists in alkaline earth metals as their outer most electron enter the S orbital and they have only two electrons in their valence shell
Q10. The paired electron can get excited and on returning back to ground state it emits light thus imparts color
Answer : Option B
Explaination / Solution:
Diamagnetic substances have full filled electrons so they are replealed by Magnetic field.
