Chemistry - Online Test
Q1. Zantac, Cimetidine, Ranitidine are all
Answer : Option C
Explaination / Solution:
They all are antihistamines.
Q2. Which of the following is non – ionizable?
Answer : Option A
Explaination / Solution:
In A there is no ion present outside the coordination sphere so it is ionizable.
Q3. Conductivity always decreases with decrease in concentration both, for weak and strong electrolytes because of the fact that
Answer : Option C
Explaination / Solution:
Conductivity of solution decreases with dilution because number of ions per unit volume decreases.
Q4. H.O.H bond angle in water is
Answer : Option C
Explaination / Solution:
Due to presence of two lone pairs on O in H2O bond angle reduce to 104.50 from 1090.
Q5. The general electronic configuration of alkali metals is
Answer : Option D
Explaination / Solution:
The general electronic configuration of alkali metals is ns1 as their outer most electron enters the S orbital and they have only on electon in their velence shell.
Q6.
Straight or branched chain compounds, for example, the below ones are called
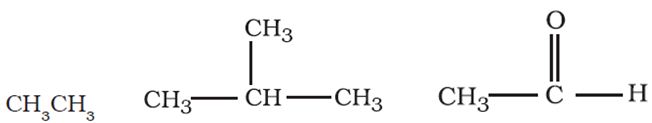
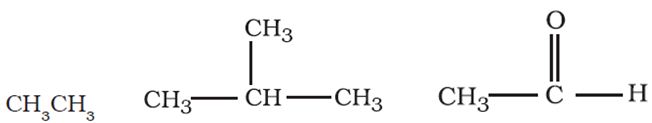
Answer : Option C
Explaination / Solution:
They are straight chain compounds known as aliphatic comounds.
Q7. According to Avogadro Law equal volumes ofall gases at the same temperature and pressure should contain ---------- number of molecules .
Answer : Option C
Explaination / Solution:
Avogadro's law states that " equal volumes of all gases at same temperature and pressure contain equal number of molecules. "
Q8. Which compound in the following pair reacts faster in SN2 reaction with OH– ?
i) CH3Br or CH3
ii) CH3Cl, (CH3)3CCl
i) CH3Br or CH3
ii) CH3Cl, (CH3)3CCl
Answer : Option A
Explaination / Solution:
For SN2 reaction leaving group should be good and alkyl halide should be primary.so CH3Br and CH3Cl are 10organic compounds hence rate of sn2 reaction will be more.
Q9. The chemical reaction in which reactants require high amount of activation energy are generally
Answer : Option A
Explaination / Solution:
If activation energy is high rate of reaction will decrease and reaction will be slow.
Q10. Liquid – liquid solutions are known as
Answer : Option D
Explaination / Solution:
Dispersed phase and dispersion medium both are liquids.
