Chemistry - Online Test
Q1. Polymerisation can be obtained by
Answer : Option A
Explaination / Solution:
Polymerisation involves Chain growth Mechanism and also condensation.
Q2. What is the spontaneous ignition temperature of white phosphorous?
Answer : Option A
Explaination / Solution:
Ignition temperature of white phosphorus is 35 oC
Q3. Standard enthalpy of fusion or molar enthalpy of fusion is
Answer : Option B
Explaination / Solution:
The enthalpy change that accompanies melting of one mole of a solid substance in standard state is enthalpy of fusion.
Q4. Which process is usually applied for separating gangue from sulphide ores?
Answer : Option A
Explaination / Solution:
Sulphide ores are concentrated using Froth floatation.
Q5. Electronic configuration of an element is:
Answer : Option A
Explaination / Solution:
The electronic configuration of an atom, describes the arrangement of electrons in space around the nucleus. The electrons are distributed over different energy level. These energy levels are called the shell or orbits.
Q6. Consider the ground state of Cr atom (Z = 24). The numbers of electrons with the azimuthal quantum numbers, l = 1 and 2 are, respectively:
Answer : Option D
Explaination / Solution:
Azimuthal quantum number l = 1 is for p and l = 2 is for d.
Now Cr has configuration
Hence there are 12, p-electrons and 5, d-electrons
Q7. Carbon monoxide is poisonous to human beings because :
Answer : Option C
Explaination / Solution:
CO has more affinity to bind to Hb than oxygen. So it blocks the binding of oxygen to Hb causing deficiency of it and can lead to death.
Q8. Zinc carbonate is precipitated from zinc sulphate solution by the addition of
Answer : Option C
Explaination / Solution:
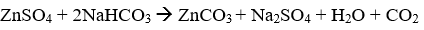
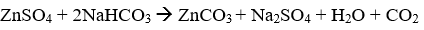
Q9. The oxidation of toluene to benzoic acid can be stopped at the aldehyde stage. The reaction is called
Answer : Option A
Explaination / Solution:
Etard is specific reaction for conversion of toluene to Benzaldehyde.
Q10. Most of the s-block elements form dihydrogen compounds which are:
Answer : Option B
Explaination / Solution:
Ionic hydrides are formed when hydrogen molecule reacts with most s-block elements which are highly electropositive in nature.
In solid state, the ionic hydrides are crystalline, non-conducting and non-volatile.
However, in liquid state it conducts electricity. Ionic hydrides on electrolysis liberate hydrogen gas at the anode.
