Chemistry - Online Test
Q1. Choose the most appropriate among the following for Bakelite
Answer : Option A
Explaination / Solution:
Bakelite undergoes extensive cross linking.
Q2. At what temperature white phosphorous changes to red phosphorous?
Answer : Option D
Explaination / Solution:
White phosphorus when heated to 573 K in an inert atmosphere for several days red Phosphorus is obtained.
Q3. The enthalpy change of a chemical reaction equals ( and are stoichiometric coefficients)
Answer : Option A
Explaination / Solution:


Q4. The diagram shows schematic representation of
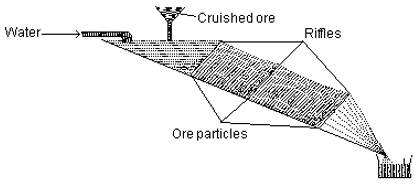

Answer : Option D
Explaination / Solution:
This is hydraulic washing or gravity separation. Here whwn stream of water is passed it takes away all the impurity with it.
Q5. According to IUPAC nomenclature,element Ununnillium have an atomic number of:
Answer : Option A
Explaination / Solution:
un=1 nil=0 So 110 = ununnilium.
Q6. When an electric discharge is passed through gaseous hydrogen, the H2 molecules dissociate and the energetically excited hydrogen atoms produced emit electromagnetic radiation of discrete frequencies. The hydrogen spectrum consists of
Answer : Option B
Explaination / Solution:
The emission spectrum of hydrogen consists of several series of sharp emission lines in the ultraviolet (Lyman series). in the visible (Balmer series). and in the infrared (Paschen series, Brackett series, etc,) regions of the spectrum. These series are named after their discoverer.
Q7. Carbon monoxide is one of the most dangerous air pollutants and is produced as a result of :
Answer : Option D
Explaination / Solution:
Carbon monoxide is produced by incomplete combustion of carbon, firewood, petrol.
The reaction is as follows :
2C + O2 -----> 2CO
Q8. Maximum oxidation state is shown by
Answer : Option D
Explaination / Solution:
Os shows maximum oxidation state of +8.
Q9. Aldehydes are prepared by reducing nitriles to corresponding imines with stannous chloride in the presence of hydrochloric acid. This reaction is called
Answer : Option D
Explaination / Solution:
Reaction for preparation of aldehydes by reducing RCN is stephens Reaction.
Q10. hydrogen compounds of non-metals are volatile compounds because
Answer : Option C
Explaination / Solution:
Covalent hydrides :They are formed when hydrogen molecule reacts with the p block elements. The most common examples, in this case, are CH4 and NH3. The hydrogen compounds formed with non-metals are also called hydrides. They are covalent and volatile compounds.
