Chemistry - Online Test
Q1. Decomposition of H2O2 can be prevented in the presence of
Answer : Option D
Explaination / Solution:
Glycerol prevents the decomposition of H2O2
Q2. Avogadro law states that
Answer : Option B
Explaination / Solution:
Equal volumes of all gases under the same conditions of temperature and pressure contain equal number of molecules. This is Avagadros Law.
Q3. For liquid ⇌⇌ vapour equilibrium, at a given temperature, the constant is
Answer : Option B
Explaination / Solution:
The vapour pressure is constant at a given temperature for liquid ⇌ vapour equilibrium. The equilibrium vapour pressure of a compound at a given temperature is an intrinsic property of the compound, meaning it does not depend on variables like volume, quantity of the compound, etc.
Q4. Glucose does not give Schiff’s test because of the formation of cyclic
Answer : Option A
Explaination / Solution:
Schiffs test is given by aldehydes, as aldehydic group is not free here.
Q5. Aldehydes are reduced to the corresponding alcohols by addition of hydrogen in the presence of catalysts to form
Answer : Option C
Explaination / Solution:
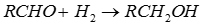
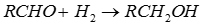
- Aldehydes and ketones are most readily reduced with hydride reagents.
- The reducing agents LiAlH4 and NaBH4 act as a source of 4 x H- (hydride ion)
- Overall 2 H atoms are added across the C=O to give H-C-O-H
- Hydride reacts with the carbonyl group, C=O, in aldehydes or ketones to give alcohols.
- The substituents on the carbonyl dictate the nature of the product alcohol.
- Reduction of methanal (formaldehyde) gives methanol.
- Reduction of other aldehydes gives primary alcohols.
- Reduction of ketones gives secondary alcohols.
Q6. p – toluenesulphonyl chloride does not react with
Answer : Option C
Explaination / Solution:
does not react with all these amines
Q7. In the alkene having IUPAC name 3-Methyl-1-butene the double bond lies on carbon atom numbered
Answer : Option B
Explaination / Solution:
‘1’ is written before butene which indicates that the position of the double bond is on carbon 1.The structure of the compound is: CH2=CHCH(CH3)CH3
Q8. At constant temperature the osmotic pressure of a solution is
Answer : Option B
Explaination / Solution:
Л=cRT
Q9. Formation of coloured solution is possible when metal ion in the compound contains
Answer : Option A
Explaination / Solution:
Color in complex arises due to the presence of unpaired electrons in the system when complex reaction takes place.
Q10. Which is true for F – centers?
Answer : Option B
Explaination / Solution:
The paired electron can get excited and on returning back to ground state it emits light thus imparts color
