Chemistry - Online Test
Q1. Lines in the hydrogen spectrum which appear in the visible region of the electromagnetic Spectrum, then they are called as
Answer : Option C
Explaination / Solution:
The Balmer series or Balmer lines in atomic physics, is the designation of one of a set of six named series describing the spectral line emissions of the hydrogen atom. The Balmer series is calculated using the Balmer formula, an empirical equation discovered by Johann Balmer in 1885 Balmar series lies in visible region of Electromagnetic spectrum.
Q2. Nitrous oxide is evolved when Zn is treated with
Answer : Option D
Explaination / Solution:
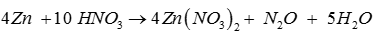
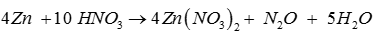
Q3. Syngas is mixture of
Answer : Option C
Explaination / Solution:
Synthesis gas or briefly, syngas, is a mixture of carbon monoxide and hydrogen. Syngas can be produced from many sources, including natural gas, coal, biomass, or virtually any hydrocarbon feedstock, by reaction with steam or oxygen. Syngas is a crucial intermediate resource for production of hydrogen, ammonia, methanol, and synthetic hydrocarbon fuels
Q4. Which of the following cation does not form an ammine complex with excess of ammonia?
Answer : Option B
Explaination / Solution:
Na+ is s block element so doesnot form ammine complex.
Q5. Which sequence of steps below describes the best synthesis of 5 – oxohexanoic acid starting with 1 – methylcyclopentan – 1 – ol
Answer : Option B
Explaination / Solution:
5 – oxohexanoic acid starting with 1 – methylcyclopentan – 1 – ol can be synthesized using conc H2SO4 which will cause dehydration forming alkene which with KMnO4 opens the ring and forms 1 – methylcyclopentan – 1 – ol.
Q6. Effluents of washing may cause pollution, if
Answer : Option D
Explaination / Solution:
If highly branched detergents molecules are present then it may cause pollution.
Q7. An increase in equivalent conductance of a strong electrolyte with dilution is mainly due to
Answer : Option C
Explaination / Solution:
Equivalent conductance increases on dilution for a strong electrolyte because of increase in mobility of ions.
Q8. Calculate the heat and the work associated with a process in which 5.00 mol of gas expands reversibly at constant temperature T = 298 K from a pressure of 10.00 to 1.00 atm
Answer : Option B
Explaination / Solution:
for isothermal process ΔU=0 while –w=nRTln(P1/P2) = q
Q9.
Based on VSEPR theory, the number of 90 degree F — Br — F angles in is
Answer : Option D
Explaination / Solution:
Steric no. of BrF5 is 6 so geometry is octahedral. It will have 5 bps and one lp which is present on axial position. Because of distortion caused by lp there is no 90o F—Br—F bond angle in BrF5 .
Q10. Which branched chain isomer of the hydrocarbon with molecular mass 72u gives only one isomer of mono substituted alkyl halide?
Answer : Option D
Explaination / Solution:
Neopentane has all same type of hydrogen and has molecular weight 72u
