Chemistry - Online Test
For the reaction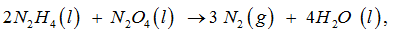
Which is the limiting reagent for this reaction ?
Chemical reactions rarely occur completely when exactly the right amounts of reactants are taken to react together to yield products as per its stoichiometry .One of the reactants will be used up ,before another runs out.Thus , the reactant which is consumed first is known as limiting reagent
For the given reaction ,
2 N2 H4 (l) + N2 O4 (l) ------> 3 N2 (g) + 4 H2 O (l)
as per its stoichiometry , if only the right and exact amount of N2 O4 is taken , the reaction may not go to completion .As such it would be consumed first & is , therefore , the limiting reagent.
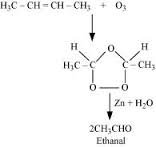
Ozonolysis of alkenes involves the addition of ozone molecule to alkene to form an unstable ozonide , and then cleavage of the ozonide by to smaller molecules .
2- butene is a symmetrical alkene , which on ozonolysis yields two moles of having molecular mass of 44 u. as per reaction given below,