Chemistry - Online Test
Q1. Heating a pulverised mixture of limestone and clay in a rotary kiln is used in the manufacture of:
Answer : Option D
Explaination / Solution:
Heating a pulverised mixture of limestone and clay in a rotary kiln is used in the manufacture of Portland cement
Q2. Covalent bond can undergo fission in two different ways. The correct representation involving a heterolytic fission of is
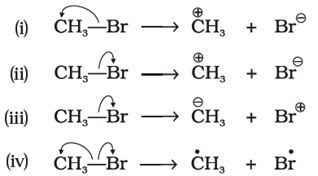
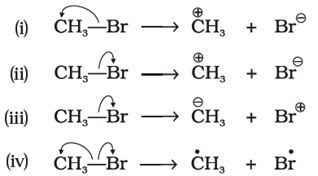
Answer : Option D
Explaination / Solution:
arrows are shown in the direction of movement of electrons.
Q3. The half life of a reaction is halved as the initial concentration of the reactant is doubled. The order of the reaction is
Answer : Option B
Explaination / Solution:
For second order reaction half life is inversely related to concentration of reactant.
Q4. Enzymes are
Answer : Option B
Explaination / Solution:
Enzymes are biological catalyst, they catalize different chemical reactions in body.
Q5. The vapor pressure of different substances at the same temperature increases if
Answer : Option A
Explaination / Solution:
if the intermolecular forces of the substance are weak then vapour pressure increases.
Q6. An example of antibody is
Answer : Option B
Explaination / Solution:
Antibody gamma globulin
Q7. Phenol is less acidic than ______________.
Answer : Option A
Explaination / Solution:
The nitro-group is an electron-withdrawing group. The presence of this group in the ortho or para position decreases the electron density in the OH bond. As a result, it is easier to lose a proton. Also, the o-nitrophenoxide or p-nitrophenoxide ion formed after the loss of protons is stabilized by resonance. Hence, ortho and para nitrophenols are stronger acids than phenol.
Q8. In which unit, the concentration of solution remains independent of temperature
Answer : Option C
Explaination / Solution:
It is independent of volume hence independent of Temperature.
Q9. The reaction of Benzene diazonium chloride with aniline yields
Answer : Option A
Explaination / Solution:
This is also N-N coupling.
Q10. How many atoms of hydrogen are in 67.2 L of H2 at STP?
Answer : Option A
Explaination / Solution:
Step 1
Number of moles of H2 in 67.2 L of H2
=
=3 moles
Step 2 /
Number of molecules in 1 moles of H2
=6.02 x 1023 molecules of H2
Since H2 is a diatomic gas the number of atoms in 1 mole of H2
= ( 2 x 6.02 x 1023 )
= 12.04 x 1023 atoms of H atoms
(since , one molecule of H2 contains 2 atoms ).
Number of atoms in 3 moles of H2
= ( 3 x 12.04 x 1023 )
=3.612x 1024 atoms of H.
