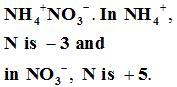Chemistry - Online Test
Q1. Liver when chronically exposed to chloroform gets damaged because
Answer : Option A
Explaination / Solution:
CHCl3 oxidises to form COCl2 which is a poison.
Q2. Calcium oxide is prepared on a commercial scale by using
Answer : Option C
Explaination / Solution:
Lime stone and rotary kiln is used in prepation of CaO.
Q3. In general, in the molecules containing multiple bonds,_____ provide the most reactive centres. Choose the most appropriate one.
Answer : Option A
Explaination / Solution:
pie bonds are the most reactive centers.
Q4. Which among the following is an instantaneous reaction?
Answer : Option A
Explaination / Solution:
Precipitation of AgCl is an example of instantaneous reaction.
Q5. In aerosol the dispersion medium is
Answer : Option C
Explaination / Solution:
Dispersion medium is gas.
Q6. A mixture of dihydrogen and dioxygen at one bar pressure contains 20% by weight of dihydrogen. Calculate the partial pressure of dihydrogen.
Answer : Option C
Explaination / Solution:
P=P1+P2 (Daltons Law of partial pressure).
Q7. The carbohydrate which cannot be hydrolysed in human digestive system is.
Answer : Option D
Explaination / Solution:
Cellulose which cannot be hydrolysed in human digestive system
Q8.
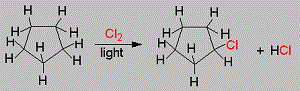
A hydrocarbon C5H10 does not react with chlorine in dark but gives a single monochloro compound C5H9Cl in bright sunlight. The hydrocarbon is
Answer : Option A
Explaination / Solution:
Cyclopentane is nearly inert chemically, they react with halogens in the presence of light through the substitution of one hydrogen atoms. Since the cyclic structure confers a high degree of symmetry on the molecule, only one monochloro cyclopentane is possible.
Q9. Solution of hydrogen in palladium is an example of
Answer : Option A
Explaination / Solution:
Hydrogen (solute , gas ) and solvent is palladium (solid).
Q10. In which of the following compounds, an element exhibits two different oxidation states.
Answer : Option B
Explaination / Solution: