Chemistry - Online Test
Q1.
A hydrocarbon C5H10 does not react with chlorine in dark but gives a single monochloro compound
C5H9C l in bright sunlight. The hydrocarbon is
Answer : Option D
Explaination / Solution:
Alkanes undergoes halogenations in prrsence of light.
Q2. Why does the solubility of alkaline earth metal carbonates and sulphates in water decrease down the group?
Answer : Option C
Explaination / Solution:
Solubility of sulphates and carbonates decreases down the group because of decrease in hydration energy. which is insufficient to over come the lattice energy of ions.
Q3. Electrophilic addition reactions proceed in two steps. The first step involves the addition of an electrophile. Name the type of intermediate formed in the first step of the following addition reaction.
Answer : Option B
Explaination / Solution:
A secondary carbocation is formed.
Q4. The half life periods of a reaction at initial concentration 0.1 mol/L and 0.5 mol/L are 200 s and 40 s respectively. The order of the reaction is
Answer : Option D
Explaination / Solution:
As initial concentration is increased half life is decreasing so order of reaction is 2.
Q5. Flocculation value is expressed in terms of
Answer : Option A
Explaination / Solution:
Fact
Q6. 2.9 g of a gas at 95 occupied the same volume as 0.184 g of dihydrogen at 17 , at the same pressure. What is the molar mass of the gas?
Answer : Option A
Explaination / Solution:
PV=(/M)R where m 1 =2.9g and =368K PV= (/2)where =0.184 and = 290K
Q7. Which one is not the essential amino acid in the ones given below?
Answer : Option C
Explaination / Solution:
Leucine is not essential amino acid
Q8. Which of the following species can act as the strongest base?
Answer : Option B
Explaination / Solution:
Hydroxy group is more electron donating group than alkoxy because alkoxy have greater number of atoms than that hydroxyl group there electronic density of oxygen of alkoxy group spreads over the whole group and greater the number of atom less will be the electron donating ability.
Q9.
Give IUPAC name of the compound given below.
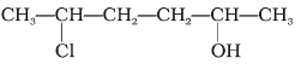
Answer : Option A
Explaination / Solution:
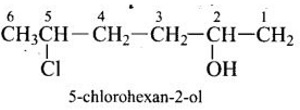
-OH is given preference over -Cl so numbering is done so that –OH gets the lowest number.
Q10. A solution showing a large positive deviation from ideal behaviour has
Answer : Option C
Explaination / Solution:
A solution showing a large positive deviation from ideal behaviour shows lower boiling point than both the components.
