Chemistry - Online Test
Q1. Lines in the hydrogen spectrum which appear in the ultraviolet region of the electromagnetic spectrum, then they are called as
Answer : Option A
Explaination / Solution:
The Lyman series is a hydrogen spectral series of transitions and resulting ultraviolet emission lines of the hydrogen atom as an electron goes from n ≥ 2 to n = 1 (where n is the principal quantum number), the lowest energy level of the electron.
Q2. Carbon dioxide and oxygen levels are maintained by:
Answer : Option D
Explaination / Solution:
Carbon dioxide and oxygen levels are maintained by Photosynthesis by plants and respiration by organisms.
Q3. Which is least soluble in water?
Answer : Option C
Explaination / Solution:
Ag2S is least soluble in water.
Q4. Calculate the strength of 5 volume solution.
Answer : Option B
Explaination / Solution:
Volume strength = 11.2 x M
So M == 0.446
Strength (g/L ) = 0.446 x 34 = 15.17 g/L
Q5. In Hell – Volhard Zelinsky reaction, the reagents used are
Answer : Option C
Explaination / Solution:
In HVZ we use X2 in presence of Red P4 .
Q6. Which of the following complexes does not obey the EAN rule?
Answer : Option C
Explaination / Solution:
23+12=35 EAN is not followed.
Q7. A patient suffering from sleeplessness was advised to take one of the following medicine in small dosages. Which is it?
Answer : Option A
Explaination / Solution:
Valium is used treat sleeplessness problem.
Q8. Resistance of 0.2 M solution of an electrolyte is 50 Ω. The specific conductance of the solution is 1.3 S . If resistance of the 0.4 M solution of the same electrolyte is 260 Ω, its molar conductivity is
Answer : Option C
Explaination / Solution:
κ= G × cell constant and G= 1/R.
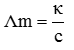
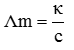
Q9.
for the reaction . Calculate for the reaction, and predict whether the reaction may occur spontaneously.
Answer : Option C
Explaination / Solution:
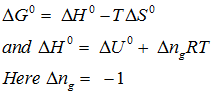
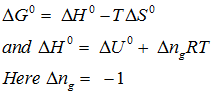
Q10. When is converted into
Answer : Option A
Explaination / Solution:
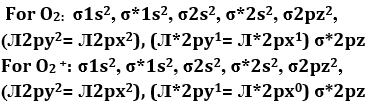
Bond order =
For O2 = = 2
For O2+ = = 2.5
