Chemistry - Online Test
Q1. An oxidation number of +2 is found in all their compounds of one of the below given options
Answer : Option D
Explaination / Solution:
Alkaline earth metals have in common an outer s- electron shell which is full; that is, that is why orbital contains its full complement of two electrons, which these elements readily lose to form cations with charge +2, and an oxidation state (oxidation number) of +2.
Q2. Which of the following is an intermediate in the mechanism for amide synthesis through acylation o
Answer : Option C
Explaination / Solution:
- A is the intermediate formed by nucleophilic attack by CH3NH2 on carbonyl carbon of CH3COCl.
Q3. For the reaction
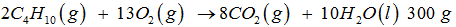
of is combusted in 1000 g of O2. The yield of and the limiting agent in the reaction are
of is combusted in 1000 g of O2. The yield of and the limiting agent in the reaction are
Answer : Option C
Explaination / Solution:
moloes of given = 51.72 mol Moles of O2 = 31.25 mol. Since 2 mol of requires 13 mol of so 51.72 mol of require 336.18 mol of but we have is only 31.25 mol therefore is limiting reagent. Therefore 13 mol of gives 10 mol of so 31.25 mol will give 24.038 mol of water = 432 g of water.
Q4. The aqueous solution of sugar does not conduct electricity. However, when sodium chloride is added to water, it conducts electricity. How will you explain this statement on the basis of ionisation and how is it affected by concentration of sodium chloride?
Answer : Option A
Explaination / Solution:
NaCl ionises completely in water and increase in concentration of salt increases conductance.
Q5. A dibromo derivative of an alkane reacts with sodium metal to form an alicyclic hydrocarbon.
The derivative is ______.
Answer : Option D
Explaination / Solution:
1,4 - dibromobutane ( ie. a dibromoderivative of an alkane , butane) when reacted withl in ethereal solution produces alicyclic hydrocarbon, Cyclobutane as per following reaction.
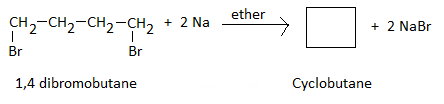
Therefore , the dibromo-derivative of alkane is identified as 1 , 4-dibromobutane.
Q6. Blister copper is about
Answer : Option D
Explaination / Solution:
Blistter Cu is 98% Cu.
Q7. Iodine is liberated from KI solution when treated with
Answer : Option D
Explaination / Solution:
Cu_2+ will reduce to Cu + and I- will oxidize to I2.
Q8. Frenkel defects are not found in alkali metal halides because
Answer : Option C
Explaination / Solution:
Alkali metals have frenkel defect because of there large size.
Q9. Hexamethylenediamine with adipic acid to manufacture Nylon 6, 6 needs the following conditions to proceed.
Answer : Option A
Explaination / Solution:
Formation of Nylon 6,6 is done at high Pressure and high temperature.
Q10. The correct increasing order of radii of following species is ----?
Answer : Option B
Explaination / Solution:
It is because
(i) radius of anion ( anionic radius ) is greater , and
(ii) radius of cation ( cationic radius ) is lesser
than that of its parent neutral atom.
