Chemistry - Online Test
Q1. Which of the following metal solution cannot be prepared by Bredig’s arc method?
Answer : Option D
Explaination / Solution:
Potassium due to very high reactivity, canot be used here.
Q2.
A student forgot to add the reaction mixture to the round bottomed flask at 27 but instead he/she placed the flask on the flame. After a lapse of time, he realized his mistake, and using a pyrometer he found the temperature of the flask was 477 . What fraction of air would have been expelled out?
Answer : Option A
Explaination / Solution:
n1/n2=T2/T1 =0.4 so fraction of air expelled out =1-0.6=0.4.
Q3. Adenosine is.
Answer : Option A
Explaination / Solution:
Adenosine is nucleoside.
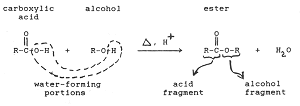
Q4. Reaction of an alcohol with organic acid is called the __________
Answer : Option C
Explaination / Solution:
Esterification is the reaction in which a Carboxylic acid combines with an alcohol in the presence of little concentrated sulphuric acid to form an ester. The esters so formed are pleasant smelling.
Q5. What will be the value of pH of
Answer : Option D
Explaination / Solution:
pH= -log(cα) And Ka=
Q6. The pH of ammonium acetate will be
Answer : Option A
Explaination / Solution:
is a salt of weak acid and weak base. pH= 7+ 0.5(pKa-pKb) pKa=-log(Ka) and pKb=-log(Kb) Here pKa=pKb so pH=7.
Q7. A solute is soluble in two immiscible liquids which are present in a mixture. The concentration of the solute in the upper layer will be
Answer : Option A
Explaination / Solution:
A solute distributes itself between two immisible liquids. Ratio of conc of solute in liquid1 and liquid 2 is constant.
Q8.
The oxidation number of hydrogen in LiH, NaH and CaH7 is
Answer : Option C
Explaination / Solution:
This is according to the rules of assigning oxidation number i.e.
Metal hydrides, such as NaH, LiH, etc., in which the oxidation state for H is -1.
Q9. When a 1° amine reacts with an alkyl sulfonyl chloride, the major organic product is __________.
Answer : Option D
Explaination / Solution:
Sulphonyl chloride reacts with primary amine to form sulphonamide.
RNH2+RI SO2Cl → RI SO2NHR + HCl
Q10. There are ___m in 2000 mm?
Answer : Option C
Explaination / Solution:
Since , 1 m
=1000 mm.
or, 1 mm
=
2000 mm
=[ ] m
= 2 m
