Chemistry - Online Test
Q1. N2O3 is
Answer : Option B
Explaination / Solution:
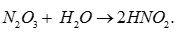
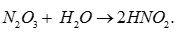
Q2. Which branched chain isomer of the hydrocarbon with molecular mass 72u gives only one isomer of mono substituted alkyl halide?
Answer : Option D
Explaination / Solution:
(CH3)3CCH3 + HX(X=Cl,Br,I) ------------> (CH3)3CCH2X
Hence a monosubstituted derivative is formed.
Q3. Which of the following metals cannot be extracted by smelting process?
Answer : Option A
Explaination / Solution:
Al cannot be extracted by smelting.
Q4.
Enthalpy of combustion of carbon to is –393.5 kJ . Calculate the heat released upon formation of 35.2 g of from carbon and dioxygen gas.
Answer : Option A
Explaination / Solution:
when 1 mole of is produced energy released is –393.5 kJ Moles of given =35.2/44 =0.8 moles So energy released = 0.8 x393.5 KJ/mol = 315 KJ/mol
Q5. Silicon carbide is an example of
Answer : Option B
Explaination / Solution:
SiC is a network solid.
Q6. The elements charecterised by the filling of 4 f-orbitals, are:
Answer : Option A
Explaination / Solution:
The two rows of elements at the bottom of the Periodic Table, called the Lanthanoids, Ce(Z = 58) – Lu(Z = 71) and Actinoids, Th(Z = 90) – Lr (Z = 103) are characterised by the outer electronic configuration (n-2)f1-14 (n-1)d0-1ns2. The last electron added to each element is filled in f- orbital. These two series of elements are hence called the Inner-Transition Elements (f-Block Elements).
Q7. In which of the following polymers ethylene glycol is one of the monomer units?
Answer : Option A
Explaination / Solution:
Formation of Dacron involves ethylene glycol and terephthalic acid as monomer unit.
Q8. Around 1015 Hz corresponds to the region of the electromagnetic spectrum
Answer : Option C
Explaination / Solution:
Electromagnetic radiation in this range of wavelengths is called visible light or simply light. A typical human eye will respond to wavelengths from about 390 to 700 nm. In terms of frequency, this corresponds to a band in the vicinity of 430–770 THz.
Q9. Unleaded paint and petrol were introduced because:
Answer : Option B
Explaination / Solution:
Unleaded paint and petrol were introduced because lead affect the brain and cause mental impairment
Q10. In dilute alkaline solution, MnO4−− changes to
