Chemistry - Online Test
Q1. Metal hydrides are ionic, covalent or molecular in nature. Among LiH, NaH, KH, RbH, CsH, the correct order of increasing ionic character is
Answer : Option B
Explaination / Solution:
As the size of cation increases ionic character also increases.
Q2. Reduction of aldehydes and ketones into hydrocarbons using zinc amalgam and conc. HCl is called
Answer : Option C
Explaination / Solution:
Clemmenson is the reaction of carbonyl compounds with Zn amalgam in presence of conc. HCl to convert them to alkanes.
Q3. The complex with highest stability constant at 298K is
Answer : Option B
Explaination / Solution:
Stability is proportional to CFSE value which is dependent upon Δo value. CN− is stronger field which increases the CFSE.
Q4. The effective medicine to be given to a person suffering from hyperacidity is
Answer : Option C
Explaination / Solution:
Cimetidine is effective in treating hyperacidity.
Q5. How much charge is required for the reduction of 1 mol of to Cu?
Answer : Option C
Explaination / Solution:
For reduction of 1 mol of to Cu, 2 mol of electrons are required so total charge will be 2F.
Q6. The number of types of bonds between two carbon atoms in calcium carbide is
Answer : Option C
Explaination / Solution:
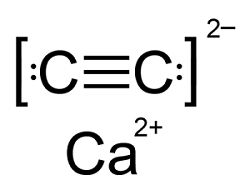
CaC2 has a combination of bonds. It is an ionic lattice that has Ca2+ cations and acetylide C22- anions. Within each C22-there is a triple bond between the 2C atoms, consisting of 1 sigma and 2 pi bonds.
Q7. Triiodomethane (Iodoform) is
Answer : Option C
Explaination / Solution:
The compound finds small scale use as a disinfectant.Around the beginning of the 20th century it was used in medicine as a healing and antiseptic dressing for wounds and sores, although this use is now superseded by superior anticeptics..
Q8. To acquire noble gas configuration, alkaline earth metals lose
Answer : Option D
Explaination / Solution:
Alkaline Earth Metals loses 2 electron to attain noble gas configuration.
Q9. To separate mixtures into their components, and also to test the purity of ompounds, the popularly used technique is :
Answer : Option A
Explaination / Solution:
Chromatography is used.
Q10. Which among the following is an example of first order reaction?
Answer : Option A
Explaination / Solution:
Decomposition of is 1st order reaction.
