Chemistry - Online Test
Q1. CH3 CHO and C6 H5 CH2 CHO can be distinguished chemically by:
Answer : Option D
Explaination / Solution:
CH3 CHO will give iodoform test and C6 H5 CH2 CHO will not give iodoform test.
Q2. In the gas phase water is a bent molecule with a bond angle of
Answer : Option B
Explaination / Solution:
Because the two non-bonding pairs remain closer to the oxygen atom, these exert a stronger repulsion against the two covalent bonding pairs, effectively pushing the two hydrogen atoms closer together. The result is a distorted tetrahedral arrangement in which the H—O—H angle is 104.5°
Q3. The element in soap which makes it soft on skin is
Answer : Option C
Explaination / Solution:
It is potassium which makes it soft on skin.
Q4. Which of the following ligand gives chelate complexes?
Answer : Option D
Explaination / Solution:
Oxalate is a bidentate ligand.
Q5. An electrochemical method is to provide a sacrificial electrode of another metal (like Mg, Zn, etc.) which corrodes itself but saves the object. A typical example is
Answer : Option C
Explaination / Solution:
Galvanization is a process in which iron is coated with reactive metal like Zn which get corroded hence preventing iron from corrosion.
Q6. The shift in electron density is symbolised by crossed arrow in the below diagram. It depicts
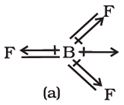

Answer : Option B
Explaination / Solution:
The cross base arrow demonstrates the net dipole. On the cross-base arrow, the cross represents the positive charge and the arrow represents the negative charge.
Q7. Hydrocarbons are prepared from Grignard reagent by
Answer : Option C
Explaination / Solution:
Grignard reagent abstracts acidic hydrogen from H2O forming alkane.
Q8. Carius tube is used in the estimations of one of the following
Answer : Option C
Explaination / Solution:
It is used in the estimation of Halogen and Sulphur.
Q9. The alkali metals dissolve in liquid ammonia giving deep blue solutions. When they become diamagnetic, the solution turns into
Answer : Option C
Explaination / Solution:
When the solution become diamagnetic due to formation of electron cluster in which ammoniated electron with opposite spin group- it changes its color to bronze.
Q10.
The relationship between temperature and rate constant is given by the relation:
Answer : Option C
Explaination / Solution:
A is Arrhenius factor.
