Chemistry - Online Test
Q1. The adsorbent used in decolouration of vinegar and sugar solution is
Answer : Option C
Explaination / Solution:
It adsorbs the colour in solutions.
Q2. The three states of matter of are in equilibrium at
Answer : Option A
Explaination / Solution:
Triple point is the point where all the three states coexist in equilibrium.
Q3. Hydronium ion concentration in molarity is more conveniently expressed on a logarithmic scale known as the
Answer : Option A
Explaination / Solution:
pH= -log[]
Q4. In aqueous solution, amino acids behave like:
Answer : Option C
Explaination / Solution:
As both acidic and basic group is present and zwittter ion is formed
Q5. In the reaction
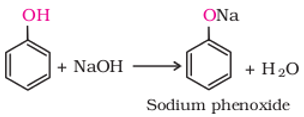 `
`
 `
`
Answer : Option A
Explaination / Solution:
Phenol reacts with sodium hydroxide solution to give a colourless solution containing sodium phenoxide. In this reaction, the hydrogen ion has been removed by the strongly basic hydroxide ion in the sodium hydroxide solution.
Q6. Aromatic compounds are charachterized by the presence of _____ π electrons in the ring where n is an integer (n = 0, 1, 2, . . .).
Answer : Option D
Explaination / Solution:
Huckel rule which explains the unusual stability of cyclic conjugated polyenes(i.e. aromatic compounds) over other cyclic unsaturatrd compounds which do not contain (4n+2) pi electrons.
Q7. Solution is defined as
Answer : Option A
Explaination / Solution:
Solution is Homogeneous mixture of Solute and Solvent.
Q8. Which of the following amines is most soluble in water?
Answer : Option C
Explaination / Solution:
Because of hydrogen bonding with water, ethylamine is highly soluble in water.
Q9. Oxidation number of −1/2 is assigned to oxygen atom in
Answer : Option B
Explaination / Solution:
In the superoxide ion, O2−, the oxygen has an oxidation number of −1/2. The stability of metal superoxides depends on the size and the electropositive character of the metal. The larger the metal and the more electropositive it is, the greater the stability of its superoxide.
Q10. Intermolecular forces of thermoplastic polymers are
Answer : Option B
Explaination / Solution:
Intermolecular forces are intermediate between elastomers and fibres.
