Chemistry - Online Test
Q1.
In the following reaction, the polymer formed is ?
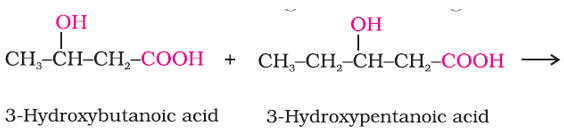
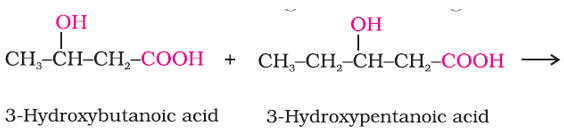
Answer : Option B
Explaination / Solution:
These monomers form PHBV.
Q2. Which of the following is the strongest Lewis base?
Answer : Option A
Explaination / Solution:
Because of lower electronegativity of I
Q3. Substances that are strongly attracted by applied magnetic field and can be permanently magnetized are
Answer : Option C
Explaination / Solution:
Ferromagnetic substance get permanently magnetised in applied Magnetic Field.
Q4. We can calculate the change in entropy of a reversible process by
Answer : Option B
Explaination / Solution:
is used to measure entropy system undergoin reversible
Q5. Ultrapurification method is used for
Answer : Option A
Explaination / Solution:
Fact
Q6. Which of the following statements is false?
Answer : Option B
Explaination / Solution:
The elements of Group 1 (alkali metals) have ns1 as outermost electronic configuration. They are all reactive metals with low ionization enthalpies. They lose the outermost electron readily to form 1+ ion. The compounds of the alkali, with the exception of those of lithium are predominantly ionic.
Q7. The Aufbau principle states : In the ground state of the atoms, the orbitals are filled in order of
Answer : Option D
Explaination / Solution:
The Aufbau Principle states that in the ground state of an atom, an electron enters the orbital with lowest energy first and subsequent electrons are fed in the order of increasing energies. The word 'aufbau' in German means 'building up'. Here, it refers to the filling up of orbitals with electrons.
Q8. Measure of the amount of organic material in the water, in terms of how much oxygen will be required to break it down biologically is termed as :
Answer : Option C
Explaination / Solution:
Measure of the amount of organic material in the water, in terms of how much oxygen will be required to break it down biologically is termed as Biochemical Oxygen Demand (BOD)
Q9. What is the shape of Fe(CO)5 molecule?
Answer : Option A
Explaination / Solution:
CN is 5.
Q10. Choose the most appropriate answer amongst the options given below for the statement -
" A solution of a desired concentration is prepared by diluting --------------- ."
Answer : Option D
Explaination / Solution:
Stock solution can best be described as concentrated solution of known accurate concentration that will be used for future laboratory use.
Since large amounts of solutes are used for preparing stock solution a more accurate concentration of it can be achieved quite easily, and as such the chances are slim to get erroneous results of the related experiments..
In addition , stock solutions are generally more stable as compared to working solution since they usually do not suppport bacterial growth.
A multiple working solution can be prepaed by dilution of stock solution using easy calculation and process.
