Chemistry - Online Test
Q1. Caustic soda is
Answer : Option A
Explaination / Solution:
Caustic Soda is NaOH.
Q2. In organic compounds, when homolytic cleavage takes place, one of the electrons of the shared pair in a covalent bond goes with each of the bonded atoms. This results in the formation of
Answer : Option D
Explaination / Solution:
free radicals are formed by hemolytic cleavage.
Q3. Reaction which takes place in one step is known as
Answer : Option C
Explaination / Solution:
Elementary reaction are one step reactions.
Q4. Which adsorption takes place at low temperature?
Answer : Option D
Explaination / Solution:
Physical adsorption is favoured at low temperature because it involves only vanderwall interactions between adsorbate and adsorbent.
Q5. What will be the pressure of the gaseous mixture when 0.5 L of at 0.8 bar and 2.0 L of dioxygen at 0.7 bar are introduced in a 1L vessel at?
Answer : Option C
Explaination / Solution:
x1=0.8x0.5 x 1=0.7x2 And P=+
Q6. The sugar constituent of DNA is.
Answer : Option A
Explaination / Solution:
Sugar constituent of DNA is D-2-deoxy ribose.
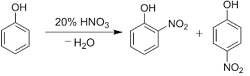
Q7. With dilute nitric acid at low temperature (298 K),phenol yields
Answer : Option D
Explaination / Solution:
Nitration of phenols: Phenols upon treatment with dilute nitric acid undergoes nitration at low temperature (298 K) to give a mixture of ortho and para nitrophenols. The mixture formed is further separated into ortho and para nitrophenols by steam distillation on the basis of their volatility. Due to intramolecular and intermolecular hydrogen bonding, ortho nitrophenols are lesser volatile in comparison to para nitrophenols which involves only intermolecular hydrogen bonding.
Q8. and are the respective ionisation constants for the following reactions.
The correct relationship between and is
The correct relationship between and is
Answer : Option B
Explaination / Solution:
Equation (3) =(1) + (2) So = ×
Q9. Which among the following is soluble in n-octane?
Answer : Option B
Explaination / Solution:
Like dissolves in like
Q10. When pentanal reacts with ethylamine under conditions of acid catalysis, the major organic product is __________.
Answer : Option C
Explaination / Solution:
Imines are typically prepared by the condensation of primary amines and aldehydes and less commonly ketones:
- RNH2 + R'C(O)R → RN=C(R')(R) + H2O
