Chemistry - Online Test
Q1. The mass of an electron is
Answer : Option C
Explaination / Solution:
The electron is a subatomic particle, symbol e− or β− , with a negative elementary electric charge.
The electron has a mass that is approximately that of the proton.
So mass of electron =
Q2. The death of Scandinavian forests was blamed on:
Answer : Option C
Explaination / Solution:
The death of Scandinavian forests was blamed on Pollution from UK power stations
Q3. How many d – block elements have ability to evolve hydrogen gas from 2% nitric acid?
Answer : Option A
Explaination / Solution:
Both Zn and Fe can displsce Hydrogen from HNO3.
Q4. Pure de-mineralised water is obtained by passing water through an anion exchange as a second process after passing it through a cation exchange. This makes the water
Answer : Option C
Explaination / Solution:
Cation exchange resin have exchangeable hydrogen ions which makes the water acidic while anion exchange resin have exchangeable hydroxide ion which makes the water basic. Passing water to anion exchange resin as a second process after passing through anion exchange resin makes the water neutral.
Q5. The compound formed as a result of oxidation of ethyl benzene by KMnO4 is
Answer : Option B
Explaination / Solution:
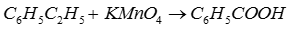
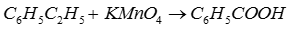
Q6. The correct name of the compound [Cu(NH3)4](NO3)2 is
Answer : Option C
Explaination / Solution:
Tetraamminecopper(II)nitrate is [Cu(NH3)4](NO3)2
Q7. The disadvantage of using Penicillin is
Answer : Option A
Explaination / Solution:
Pencillin causes allergic reactions.
Q8. The resistance of a conductivity cell containing 0.001M KCl solution at 298 K is 1500 Ω. What is the cell constant if conductivity of 0.001M KCl solution at 298 K is 0.146 × 10–3 S .
Answer : Option D
Explaination / Solution:
Κ= G x cell constant and G= 1/R.
Q9. During the formation of a chemical bond
Answer : Option C
Explaination / Solution:
A chemical bond is a lasting attraction between atoms that enables the formation of chemical compounds. The bond may result from the electrostatic force of attraction between atoms with opposite charges, or through the sharing of electrons as in the covalent bonds.
When a bond forms, electrons are attracted to the space between nuclei where the electrostatic force of attraction is greater. As the electrons fall to a position of lower potential energy, the total mechanical energy of the molecular system decreases. Part of the mechanical energy of the unbound atoms is lost when they form the molecular system with a lower total mechanical energy.
Lower the energy higher the stability of bond.
Q10. Toluene reacts with a halogen in the presence of iron (III) chloride giving ortho and para halo compounds. The reaction is
Answer : Option C
Explaination / Solution:
This is example of electrophilic aromatic substitution (EAS) reaction.Sincle Cl ion replaces hydrogen atom and here FeCl3 acts as halogen carriers.
