Chemistry - Online Test
Q1. For dissolution of solids in liquids, at a given temperature, the constant is
Answer : Option D
Explaination / Solution:
This is considered at equilibrium process.
Q2. Glycosidiclinkage:is found between
Answer : Option C
Explaination / Solution:
Glycosidic bond is an ether linkage between two monosaccharides.
Q3. Alcohols have high boiling points because of
Answer : Option C
Explaination / Solution:
In alkanes, the only intermolecular forces are van der Waals dispersion forces. Hydrogen bonds are much stronger than these and therefore it takes more energy to separate alcohol molecules than it does to separate alkane molecules. That's the main reason that the boiling points are higher.
Q4. Carbylamine reaction is one of the following in the presence of heat
Answer : Option D
Explaination / Solution:
In carbylamines reaction primary amine reacts with chloroform and base to form isocyanide having foul smell.
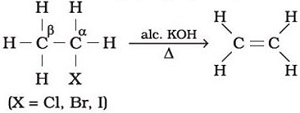
Q5.
One of the methods of preparing ethylene is given below.This is an example of _______ reaction
Answer : Option B
Explaination / Solution:
Dehydration of alcohols in the presence of alc. KOH to form an alkene is an example of elimination reaction.
Q6. The rate at which a solid dissolves in liquid does not depend on
Answer : Option B
Explaination / Solution:
The rate at which a solid dissolves in liquid does not depend on pressure.
Q7. Standard electrode potential of three metals X, Y and Z are –1.2 V, +0.5 V and –3.0 V respectively. The reducing power of these metals will be
Answer : Option A
Explaination / Solution:
X=-1.2V , Y=+0.5V , Z=-3.0V Therefore, Z>X>Y
Because, higher the reduction potential ,lesser the reducing power.
Q8. The alkali metals and their salts impart characteristic colour to an oxidizing flame because:
Answer : Option C
Explaination / Solution:
Alkali metals have one unpaired electron in their valence shell which gets excited by absorbing the energy from flame, excited states are very unstable and quickly move back to lower state ,at that time they emit the energy in the form of radiation in visible reason of electromagnetic spectrum and is abserved as characteristic colour.
Q9. All those atoms or molecules which have an odd number of electrons are
Answer : Option D
Explaination / Solution:
They will have unpaired electrons
Q10. Which polymers occur naturally in nature
Answer : Option C
Explaination / Solution:
Starch and cellulose occur naturally.